by All Things Neonatal | May 30, 2015 | bioethics, Neonatal, Pregnancy
A couple months ago on my Facebook Page (www.facebook.com/AllThingsNeonatal) I posted the shocking story of a 65 Grandmother Annegret Raunigk who received IVF and was carrying quadruplets. The post spawned outrage among my followers with statements that it was simply wrong while others argued that the risks of the mother giving birth to premature infants who would need extensive support was extremely high and that it was unethical to have done so. From my standpoint I agreed with the comments and so here you have it; the mother (or grandmother) has given birth.
http://www.cnn.com/2015/05/24/europe/germany-grandmother-quadruplets/index.html
She has delivered four premature infants < 26 weeks gestational age. Assuming they are 25 weeks each the outcomes for these babies may not be as dire as one might think. We would expect for singletons about a 70 – 80% survival with about 50% surviving without moderate or severe impairment. With quadruplets I would expect lower numbers so the reality is that at least two of these kids will have significant health care needs in the future. I would ask that we leave aside the arguments that may ensue at this point by bioethical purists who may argue that the babies’ perception of their quality of life in adolescence would be better that ours. The reality is that even if this were so, this single mother has now given birth to 17 children of which her last four are extremely low birth weight.
Who will be there to care for these children? What about the impact on society? in the next ten years will she have the energy to provide the stimulation, take the kids to all their appointments and so forth that will be needed to ensure an optimal outcome for them from a developmental standpoint?!
This is wrong on so many levels and it is not enough to say as they mention in the article that doctors encouraged her to undergo fetal reduction by one or two fetuses at an early stage. That excuses the IVF treatment in the first place by ignoring the fact that the opportunity to do something about this was missed at the first doctor’s appointment when she asked for the IVF treatment.
Nadya Suleman the “Octomom” give birth to eight premature infants and became known the world over in a sensational news story that followed her through her pregnancy and birth. It is another example of the medical community embarking on a path that lacks responsibility and ignores one of the founding principals of our Hippocratic Oath “To do no harm”. Nadya’s physician in her case suffered one of the most appropriate punishments that California could dole out. He lost his license (http://nydn.us/1d5rSxc), she went on to file bankruptcy, spoke out against the kids she wanted so much and finally resorted to pornography to pay the bills (http://huff.to/1AE5HsR). I can only wonder if the mainstream media’s obsession with Nadya’s story sparked desires for copycat sensational pregnancy stories elsewhere. Annegret Raunigk will not doubt receive a tremendous amount of attention as she already has so is there another agenda over and above to have children that is at work here?
As a medical practitioner it is generally ill advised by the local Colleges to speak out publicly against another physician but in this case this doctor who chose to implant embryos into the 65 year old mother should suffer the same fate and lose his license before he does such a thing again. He lacked judgement and any sensibility in creating a situation that would almost certainly lead to four extremely premature infants being born. I hope the German medical community acts to remove such a physician (I use the term lightly) before he can cause any further damage.
by All Things Neonatal | May 28, 2015 | Innovation, Neonatal, Neonatology
Update
Based on what you will read below our centre implemented the practice change below. In a two year span we saw our repeat sampling for metabolic screens drop from about 800 per year to 300! A small change with large benefits!
A Little Less Pain
The 1960s saw the emergence of newborn screening for phenylketonuria. This was an important milestone in the field of newborn care as it allowed us to screen children for something that we could do something about. Dietary manipulation could for the first time prevent the repercussions of this condition and allow these children to avoid the severe neurological impairment that would follow the natural course of the condition. Since that time, our ability to screen for and offer treatment to modify other disease courses has expanded many fold which no doubt in terms of population health is a wonderful thing.
Here in Manitoba we are now screening for over 40 conditions with a useful site for information being provided by Manitoba Health.
The expansion of these programs has been possible due to the use of Tandem Mass Spectrometry. This technique provides the ability to screen for many conditions without increasing the amount of blood required.
The downside to more screening is that as the number of tests being sampled increases the risk of false positive results due to the presence of dietary additives. An example of this is carnitine supplementation. In our centre we were providing this to low birth weight infants based on demonstrated low levels of carnitine facilitated lipid metabolism. After failing to find a clinical benefit after the metabolic derangements were noted we identified a larger issue in that many of our premature infants receiving carnitine supplementation had elevated acylcarnitine profiles on their Newborn Metabolic Screening (NMS) samples. These false positive results led to repeated sampling via bloodspot analysis leading to unnecessary blood sampling and pain from heel lances.
Another set of conditions that we are now able to screen for are the aminoacidopathies. This group of disorders involve abnormalities of amino acid metabolism leading to toxic elevations of one or more amino acids that can have significant neurodevelopmental impairment as a consequence. Clearly in all of these tests the purpose is to avoid long-term deleterious consequences but as with carnitine, false positive results are very concerning as they lead to repeated sampling, and potentially larger blood draws if confirmation of the screening results are needed. Add to this, that this further analysis requires consultation with metabolics consultants, nursing time for repeated sampling, and laboratory costs and you can see why minimizing false positives is needed. Lastly the greatest impact is on the family who in many cases experience unneeded anxiety as they await confirmatory testing which may take a week or more to come back if the sample needs to be sent offsite.
A few years back I attended the PAS meeting in Boston and heard about a study on this subject from California that they were presenting in abstract form. Withholding TPN and using D10W for a three hour period prior to collection of the NMS could reduce false positive aminoacidopathy screens by about 70%. The reaction of our local laboratory was one of disbelief as the consensus was that such a short time frame could not clear the TPN sufficiently from the circulation. Since the reference ranges for normal amino acid profiles in infants are from patients who are not receiving TPN this could create false positive elevations, which would require either repeat blood spot sampling or as above, trigger a formal consult to metabolics if the subsequent test is also positive.
In following up on the original abstract presentation I noted that the findings were in fact published as Reduction in Newborn Screening Metabolic False- Positive Results Following a New Collection Protocol.
In this 2 year retrospective cohort study, in 2010 NMS was done for all infants between 24-48 hours with no withholding of TPN and in 2011 the protocol was changed to hold TPN for 3 hours and use D10W before collection of the NMS. The main results of the study are shown below and of note all the False Positive results post intervention were statistically different to a significant degree after the change in practice. Examining the entire group there was a 74% reduction in false positive results post practice change.
|
Preintervention (N=274) |
Post Intervention (N=265) |
| Birth Weight |
Negative (%) |
False Positive (%) |
Negative (%) |
False Positive (%) |
| <1000g |
13 (65) |
7 (35) |
14 (82.4) |
3 (17.6) |
| 1001-1500 g |
23 (92) |
2 (8) |
24 (96) |
1 (4) |
| >1500 g |
222 (96.9) |
7 (3.06) |
223 (100) |
0 (0) |
| Total |
258 (94.2) |
16 (5.83) |
261 (98.5) |
4 (1.50) |
Furthermore an 81% savings in health care costs per patient were realized in the change as well. This is outlined below:
| Item |
Preintervention |
Postintervention |
| Confirmatory testing |
4653 |
434 |
| Supplies for testing |
46 |
12 |
| Supplies for new protocol |
0 |
465 |
| Total |
4699 |
911 |
The results speak for themselves yet the practice I don’t believe has been widely adopted and certainly not in our centre. This past week however the following study was released in abstract form and inspired me to write this post as I believe the evidence is overwhelmingly in support of this practice change Stopping Parenteral Nutrition for Three Hours Reduces False Positives In Newborn Screening.
12 567 consecutive births in 1 hospital between May 2010 and June 2013 were analyzed to determine the FP for AA levels in the NMS. The FP rate in infants > 1500g was much lower overall than for those under 1500g which may have been explained by less TPN use in that cohort. Similar to the first study TPN was changed to D10W for three hours prior to collection of the NMS and resulted in a FP rate of 3.1% in the D10W group vs 11.8% in the TPN group. This represents again an overall 74% reduction.
So there you have it. Two studies showing the same results. The concept is simple, saves hospitals money and more importantly avoids unnecessary parental anxiety, needless blood sampling and consumption of time by nursing staff and other consultants. This is not a high tech strategy that takes a great deal of education to implement. Rather this can be started tomorrow wherever you are and it is my hope that by reading this at least one hospital out there aside from our own may adopt this small change to make a big impact on our patients.
by All Things Neonatal | May 25, 2015 | bioethics, Neonatology
Virtually every popular media site and especially those dedicated to covering the world of Obstetrics and Neonatology have been ablaze with discussions on the NEJM paper Between-Hospital Variation in Treatment and Outcomes in Extremely Preterm Infants. Since this publication, numerous blogging sites have similarly expanded on the coverage with testimonials of success stories of infants born too small or too young who survived and in most cases nearly intact.
Other posts are even more sensational, claiming that the infants have done extremely well and are in fact normal and healthy. The stories capture all of our attention and as feel good pieces give us pause to reflect on the tenacity of our beliefs either for or against resuscitation of these patients. Even more powerful are those instances when our health care teams experience a good outcome at these gestational ages. These types of cases challenge even the most ardent individuals that as a rule we should not intervene before 24 weeks.
How Likely Is A Good Outcome at 22 & 23 weeks?
What we have to remember though is that at least for the 22 week infants < 10% of those reported in the US cohort survived without moderate or severe disability and in the case of the 23 week infants these numbers although better, gave a 1/6 chance of surviving without that outcome. Flipping this number on its head to present a glass is half empty argument, there is a 5/6 or an 83% chance that a child born at 23 weeks who is actively resuscitated will either die or be left with a moderate or severe disability. The blog posts from this group of parents are far less common, although I have no doubt many of these parents were happy they at least tried. Had they not they would have been left wondering for the rest of their lives “what if”?
The purpose of this post is not to debate the rightness or wrongness of the desire to resuscitate or not. Rather I would like to discuss the fallacy of thinking that informed consent plays any role in most of these cases. All too often clinicians hide behind the veil of “informed consent” as justification for the family choosing to move forward with the decision to try to resuscitate these small infants. Is that what is really happening though in most cases? Sadly, in the nearly 12 years I have been in practice in these circumstances I suspect the conditions for true informed consent have been absent more often than not. Yet the Canadian Pediatric Society lists as the third recommendation in the statement “Counselling and management for anticipated extremely preterm birth”
“Decision making between parents and health professionals should be an informed and shared process. Decision aids may be helpful for parents. (Strong Recommendation)”
The issue lies in the criteria to obtain informed consent.
The Merriam-Webster Dictionary provides this definition:
“A formal agreement that a patient signs to give permission for a medical procedure (such as surgery) after having been told about the risks, benefits, etc.”
Unless a family has previous experience with a baby born at 23 or 24 weeks (outcomes after that become quite different), or they themselves work in the field of Pediatrics and have seen the range of outcomes that these children experience, how can they really know what the risks and benefits are?
As a young medical student I recall being told time and time again to be careful about the use of medical jargon. Rather, ensure that you use lay terminology that the parents can understand, in order to ensure that informed consent can be obtained or that the family understands the clinical course of their child. How many times have I talked to families using such words of wisdom only to think in the back of my mind; “do they really understand what I am saying?”.
What do they know of chronic lung disease, PDA, IVH, ROP and a host of other problems? What can they possibly know what their lives will be like in 2, 5 or 10 years down the road should their infant survive but with a host of major medical issues? Add to this, that in many cases there is little time for discussion as the mother presents with rupture of the membranes and goes on to deliver within 48 hours of presentation and often an even shorter time frame. Furthermore, the history of the pregnancy can be extremely motivational in helping a family decide what course is right for them. How does the mother who has had three rounds of IVF perceive the situation compared to the 16 year old who had an unplanned pregnancy. Clearly the psychological factors at play must have a tremendous influence on the decision.
Many parents are simply not in a state to receive the information even if it is presented well.
A study by Boss R et al suggests that what we say has very little impact on the parental decisions (Values parents apply to decision-making regarding delivery room resuscitation for high-risk newborns). In this paper, 26 mothers responded to a questionnaire at a minimum of 10 months after the death of their infant who was born between 22 and 25 weeks. After being asked about the discussions they had with their Neonatologist and Obstetricians these were the four main points uncovered.
1. Parents rarely perceive that there are options for delivery room resuscitation. They recall being told information but rarely that compassionate care was even an option even if this was documented in the chart.
2. Physicians’s predictions of morbidity and death are not central to parental decision-making regarding delivery room resuscitation. In essence the parent’s “gut instinct” which was nearly uniformly positive guided their decision.
3. Religion spirituality and hope are the primary values that parents apply to decision-making regarding delivery room resuscitation.
4. Parents feel abandoned without physician hope and compassion. There was mistrust of doctors who only emphasized the negative without a sense of emotion. Relaying the facts is not enough.
When under such stress is it possible for families to weigh all of the true risks and benefits and come to a conclusion that satisfies the requirements for informed consent? Given that to do so requires that an individual be in a state that allows them to process this information and take their time in coming to a conclusion I think in most cases the answer is no.
Planning a Path Forward
If your centre is planning to cross over the threshold of 24 weeks there must be a coordinated plan between the delivering obstetrician, neonatal team and family. Most parents are in no position to truly appreciate what we are telling them either because of the emotional frightened state they are in or because the overpowering urge to “do everything you can” takes a front and centre position in their mind. All we can do as the Health Care providers is to provide them with as accurate information as we can and support each family after they make their decision. Once that decision is made we must also realize that once we embark on a path it is not permanent. If the condition of the newborn is such that survival without severe disability becomes increasingly unlikely we owe it to the whole team (including the family at the centre) to revisit what would be the best course of action.

by All Things Neonatal | May 22, 2015 | NAS
In medical school you learn about the concepts of primordial, primary, secondary and tertiary prevention which can be explained in the following figure.
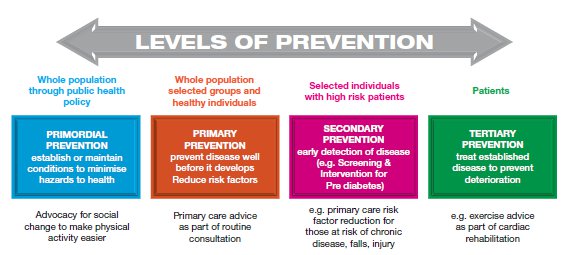
Simply put, from an impact standpoint you are far better to try to target the condition of interest with strategies to prevent the condition from happening than minimizing consequences once the condition is present in the patient.
As I wrote in another piece on Neonatal Abstinence recently we are experiencing an epidemic in mothers using opioids during pregnancy whether they be prescribed or obtained for recreational use. The US and Canada have taken great strides to enact preventative strategies to minimize exposure by introducing prescription tracking methods to monitor how often people are seeking these medications and identify those who are “doctor shopping”. While this should help deal with excessive use and furthermore curtail the selling to others who cannot be tracked, it still falls short of preventing people from using them in the first place.
We have the means to deal with the infants of these pregnancies but as mentioned in the other related piece, the number of hospital days and costs to the health care system have become staggering over time so clearly this is not the best option as we are focusing on tertiary prevention by treating the infants.
What inspired me to write this post was a publication entitled “Longitudinal cognitive development of children born to mothers with opioid and polysubstance abuse” by Nygaard E et al.
While long-term outcome studies on cocaine and alcohol as an example exist and show exposure in pregnancy can have both teratogenic and developmental affects, the impact of opioids in the long run has been less well-defined and limited to shorter lengths of outcome. An example of the effects of cocaine can be found here
Furthermore what is the impact of opioids in the presence of polysubstance use?
In the present study, the results are fairly alarming
Seventy two children with opioid and polysubstance exposure during fetal development were compared to 58 children without such exposure. Matching known risk factors for adverse outcome including such things as SES, placement in foster care was done. The results at 4 years showed similar results to other studies in that the groups exposed to the substances had lower cognitive functioning that the unexposed group with a mean difference of 10.5 IQ points at 4 years of age. What was striking though is that when the group was retested at 8.5 years of age, the gap had widened with a mean difference of 20.6 IQ points. Further analysis revealed that in fact the boys in the group had not deteriorated but it was the girls in this case that explained the difference with a mean difference of 14.4 IQ points.
The girls in this cohort showed substantial decline in cognitive abilities compared to the unexposed females.
This is the first study to show such cognitive decline over time especially in a female population. The reasons for this decline are speculative at best but may be related to different social functioning for females and males in foster care or other environments compared to males. Whatever the case, the exposure prenatally seems to have long-lasting consequences which become worse over time. In fairness I should mention that this is a small study and therefore open to risk of not representing what a larger cohort may show but the trend is certainly in keeping with other literature before it.
Coming back to the start of this piece, this publication serves as a strong reminder that if we wish to make a big difference to the greatest number of children we need to focus our efforts not on the problem in our NICUs but before the pregnancy occurs in the first place. Once the pregnancy has occurred public health campaigns could help educate women on the potential harm from using other substances when using opioids. An educational strategy is not doubt the way to go as the admission to the NICU is really the tip of the iceberg. The real cost is downstream and pertains to the societal impact, loss of function and dependency on the health care system for the rest of their lives. Lastly the children of these affected pregnancies themselves are at higher risk of repeating the cycle again enforcing the point that the time to do something is now using primordial and primary prevention.

by All Things Neonatal | May 15, 2015 | caffeine, Neonatal, Neonatology
For those of you who know me and my practice as a Neonatologist you may find the title of this piece odd. I have and will likely continue to be an advocate for the use of caffeine in premature infants. I recommend it both very early in the caseroom for those under 32 weeks to help stave off intubation and often continue caffeine until late in an infants’ stay in the NICU. Truth be told I also send children home on caffeine on occasion when all other markers needed for discharge have been met but they continue to have episodes of apnea and bradycardia that are not resolving and prolonging their stay in hospital.
In recent years I have noticed a creep of practice to begin pushing doses of caffeine base beyond the 5 mg/kg level that has been generally accepted as the upper limit of the 2.5 to 5 mg/kg range that most use in practice. The standard dosing was justified based on the CAP study by Schmidt et al indicating that it was effective in reducing the risk of bronchopulmonary dysplasia and success at earlier extubation. While there appeared to be an initial benefit to neurodevelopment favouring caffeine treatment by school age the difference disappeared. This creep effect to using higher daily maintenance dosing of 7 or 8 mg/kg/d has occurred likely for some good reasons not the least of which is a dose effect in which clinicians could see a reduction in clinical events for some patients as they increased the dose. We are no different as doctors than others in that success tends to shape our practice. Now before you accuse us of being mavericks, we did have some evidence to support the use of higher dosing beyond the 5 mg/kg dosing that had been recommended. Published in 2004, Steer and colleagues studied the effect of using a loading dose of 80 mg/kg caffeine citrate (take 50% reduction to get the base formulation we normally use) followed by 20 mg/kg maintenance dosing vs 20 mg/kg loads and 5 mg/kg maintenance in a cohort of infants < 30 weeks gestation who were having a planned extubation. The full article may be found here. The results of the study demonstrated greater success in extubation and less apnea in the group treated with the higher doses as shown here.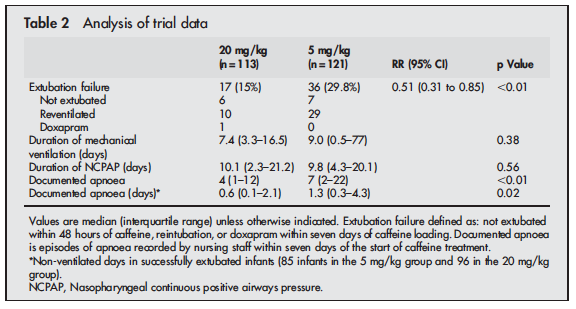
The results of this study certainly made some waves in the Neonatal community as can be seen by the “creep” in practice over the last number of years to increase the caffeine dose in our units to 6, 7 and sometimes 8 mg/kg of caffeine base in an effort to essentially titrate to effect especially in infants who are on CPAP. The motivation to prevent a reintubation secondary to apnea has been so compelling that the theoretical concerns over lack of long-term outcome data on high dose caffeine treatment have been largely ignored.
At this point it is important to also recognize that the way in which we use caffeine in terms of initiation of treatment has also changed. Many units have adopted the “Golden Hour” approach to neonatal resuscitation and are driven to use non-invasive means of support after encouraging results from several trials such as the Support, Boost and the more recent Canadian NIPPV trial. While not demonstrating improvements in outcomes necessarily, the fact that BPD rates are mostly unchanged means that with the use of early caffeine in the delivery room and the use of CPAP one can avoid invasive ventilation in many infants. As such, there has been a departure from the practice as described by Steer and colleagues to using caffeine to facilitate extubation to trying to prevent it in the first place.
In discussions with some of my colleagues we have expressed some reservation over the use of the higher doses of caffeine beyond 5 mg/kg and with the publication of a study this week by McPherson et al, these concerns may be quite warranted. For the complete study click here. This study of 74 preterm infants randomized them in the first 24 hours of life to either 80 mg/kg or 20 mg/kg caffeine citrate loads and then in both groups they followed these loads with 10 mg/kg per day maintenance. The primary outcome of the study was white matter structural development by MRI. Previous research by Doyle had found an improvement in this outcome with the use of standard caffeine therapy of 10 mg/kg/d so the real question here was “If a little is good, then is more better?”
Sadly the answer to the last question is a resounding NO!
None of the respiratory outcomes were any different between the standard caffeine and high dose groups but the following came out as a worrisome outcome:
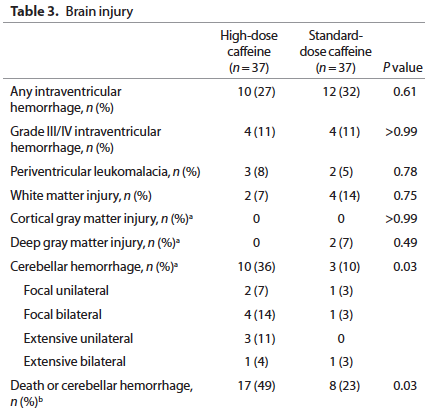
Furthermore when the infants were followed up at 2 years of age a statistically significant percentage of 2 year olds previously randomized to the high dose caffeine regimen were found to be hypertonic (2.3 vs. 1.5%). Overall neurodevelopment was no different between groups but it should be pointed out that the study was not powered to detect such differences.
One question that must come up with these findings is whether or not it is plausible that a 2 day exposure to high dose caffeine followed by standard dosing for the remainder of the time could lead to cerebellar hemorrhage. I think the answer is yes given the findings from a single dose of 25 mg/kg caffeine (equivalent to 50 mg of caffeine citrate/kg as studied by Hoecker et al
http://www.ncbi.nlm.nih.gov/pubmed/11986437
As noted by the authors, this single dose was responsible for reducing cerebral blood flow velocity by about 20% from baseline. The regimen over 48 hours in the above study was to give 80 mg/kg in divided doses as a load so it is reasonable to conclude these infants would have experienced a reduction in cerebral blood flow as well, and possibly to a greater degree than the patients in the Hoecker study. Add to this that these are infants under 30 weeks of age who have a fragile arterial and venous network to begin with and it seems reasonable that a period of hypoperfusion possibly combined with hypoxemia and then reperfusion injury could account for these cerebellar bleeds.
So where does this leave us? As the authors conclude it is not wise to plan a larger study looking at the same strategy given the findings in this pilot. What remains unclear at least to me is whether 6, 7 or 8 mg/kg during the maintenance phase of treatment offers any true long-term advantage. With anything there are tradeoffs though and finding the right balance is never easy. If we use lower caffeine doses and in some patients they require intubation, is the increased risk of CLD and possible neurodevelopmental impairment from that worth the limitation of risk? After the first week of life is the risk of cerebellar hemorrhage lower as the blood vessels mature? I think so which would make the argument for using higher doses at that point but in truth we just don’t know about safety in terms of long-term outcomes. For now at least it would seem that in the absence of guidance from research all we can really say is that 2.5 to 5 mg/kg/d of caffeine base is safe but that doses higher than that need to be used with caution. It may be wise to seek informed consent for the use of higher doses in light of these findings but it is up to each unit to decide if this is justified based on your views of the data. What do you think?








