by All Things Neonatal | Jan 15, 2018 | BPD, Neonatal, Neonatology, newborn, outcome, preemie, Prematurity, steroids
This must be one of my favourite topics as I have been following the story of early hydrocortisone to reduce BPD for quite some time. It becomes even more enticing when I have met the authors of the studies previously and can see how passionate they are about the possibilities. The PREMILOC study was covered on my site twice now, with the first post being A Shocking Change in Position. Postnatal steroids for ALL microprems? and the second reviewing the 22 month outcome afterwards /2017/05/07/early-hydrocortisone-short-term-gain-without-long-term-pain/.
The intervention here was that within 24 hours of birth babies born between 24-27 weeks gestational age were randomized to receive placebo or hydrocortisone 1 mg/kg/d divided q12h for one week followed by 0.5 mg/kg/d for three days. The primary outcome was rate of survival without BPD at 36 weeks PMA. The finding was a positive one with a 9% reduction in this outcome with the use of this strategy. Following these results were the two year follow-up which reported no evidence of harm but the planned analysis by gestational age groupings of 24-25 and 26-27 weeks was not reported at that time but it has just been released this month.
Is there a benefit?
Of the original cohort the authors are to be commended here as they were able to follow-up 93% of all infants studied at a mean age of 22 months. The methods of assessing their neurological status have been discussed previously but essentially comprised standardized questionnaires for parents, assessment tools and physical examinations.
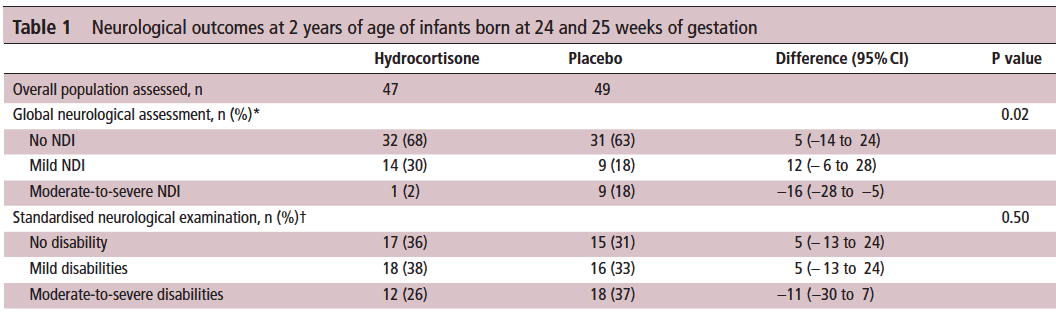
Let’s start off with what they didn’t find. There was no difference between those who received placebo vs hydrocortisone in the 26-27 week group but where it perhaps matters most there was. The infants born at 24-25 weeks are certainly some of our highest risk infants in the NICU. It is in this group that the use of hydrocortisone translated into a statistically significant reduction in the rate of neurodevelopmental impairment. The Global Neurological Assessement scores demonstrated a significant improvement in the hydrocortisone group with a p value of 0.02. Specifically moderate to severe disability was noted in 18% compared to 2% in the group receiving hydrocortisone.They did not find a difference in the neurological exam but that may reflect the lack of physical abnormalities with cognitive deficit remaining. It could also be explained perhaps by the physical examination not being sensitive enough to capture subtle differences.
Why might this be?
Adding an anti-inflammatory agent into the early phase of a preemies life might spare the brain from white matter damage. Inflammation is well known to inflict injury upon the developing brain and other organs (think BPD, ROP) so dampening these factors in the first ten days of life could bring about such results via a mechanism such as that. When you look at the original findings of the study though, a couple other factors also pop up that likely contribute to these findings as well. Infants in the hydrocortisone group had a statistical reduction in the rate of BPD and PDA ligations. Both of these outcomes have been independently linked to adverse neurodevelopmental outcome so it stands to reason that reducing each of these outcomes in the most vulnerable infants could have a benefit.
In fact when you add everything up, is there much reason not to try this approach? Ten days of hydrocortisone has now been shown to reduce BPD, decrease PDA ligations and importantly in the most vulnerable of our infants improve their developmental outcome. I think with this information at our fingertips it becomes increasingly difficult to ignore this approach. Do I think this will become adopted widely? I suspect there will be those who take the Cochrane approach to this and will ask for more well designed RCTs to be done in order to replicate these results or at least confirm a direction of effect which can then be studied as part of a systematic review. There will be those early adopters though who may well take this on. It will be interesting to see as these centres in turn report their before and after comparisons in the literature what the real world impact of this approach might be.
Stay tuned as I am sure this is not the last we will hear on this topic!
by All Things Neonatal | May 11, 2017 | kangaroo care, Neonatal, Neonatology, outcome, Parents Corner
Aside from me donning the costume in the above picture for the Kangaroo Challenge 2017 I learned something new today. Before I get into what I learned, let me say that I had the opportunity to put so many smiles on parents faces by walking around in this full body costume that I am grateful to Diane for finding this costume and Sue (you both know who you are) for purchasing it. Handing out cookies to the parents and children at the bedside and seeing them smile while knowing that they were under significant stress gave me the opportunity to interact with parents in a very different way than I am accustomed to as a Neonatologist so I am so thankful to have had that experience and yes if called upon I will do it again!
We even made the local news! CTV newscast
I posted the above picture on my Facebook page and to my surprise many of the comments led me to believe that Kangaroo Care is still something that needs a little nudging to get the word out about. I found this actually quite surprising given how immersed we are in Winnipeg with this strategy. When I think about new interventions in Neonatology it is synonymous in virtually all cases with an influx of dollars to achieve usher in the new program. Here is a program that is virtually free but only requires a commitment from families to spend the time at the bedside with their baby in the NICU.
I have been asked by many of my nursing colleagues to write something about Kangaroo care on this site and so here it is…
What is it?
You have likely heard of Kangaroo Care and you may have even seen some children receiving it in your hospital. Why is this so important?
Kangaroo Care (KC) or Skin to Skin Care (STS) is an ideal method of involving parents in the care of their premature infant. It fosters bonding between parents and their hospitalized infant, encourages the family to be with their child and thereby exposes them to other elements of neonatal care that they can take part in. While we know that many units are practising Kangaroo Care there is a big difference between having KC in your unit and doing everything you can to maximize the opportunity that your families have to participate.
There is much more to KC than simply holding a baby against your chest. For a demonstration of KC please watch the accompanying video and show it to any one in your units that may need a visual demonstration. This excellent video is from Nationwide Children’s Hospital and walks you through all of the important steps to get it right and maximize benefit.
Kangaroo Care Video
Before you reach the conclusion that KC only serves to enhance the parental experience it does so much more than that. The practice began in Bogota Columbia in 1979 in order to deal with a shortage of incubators and associated rampant hospital infections. The results of their intervention were dramatic and lead to the spread of this strategy worldwide. The person credited with helping to spread the word and establish KC as a standard of care in many NICUs is Nils Bergman and his story and commentary can be found here http://bit.ly/1cqIXlm
The effects of KC are dramatic and effective to reduce many important morbidities and conclusively has led to a reduction in death arguably the most important outcome. An analysis of effect has been the subject of several Cochrane Collaboration reviews with the most recent one being found here.
To summarize though, the use of KC or STS care has resulted in the following overall benefits to premature infants at discharge or 40 – 41 weeks’ postmenstrual age:
Reduction in
mortality (typical RR 0.68, 95% CI 0.48 to 0.96)
nosocomial infection/sepsis (typical RR 0.57, 95% CI 0.40 to 0.80)
hypothermia (typical RR 0.23, 95% CI 0.10 to 0.55)
Increase in
KMC was found to increase some measures of infant growth, breastfeeding, and mother-infant attachment
To put this in perspective, medicine is littered with great medications that never achieved such impact as simply putting your child against your chest. This is another shining example of doing more with less. This is not to say that modern medicine and technology does not have its place in the NICU but KC is simply too powerful a strategy not to use and promote routinely in the NICU.
Please join me in championing this wonderful technique and make a difference to all of our babies!
A sample of our parent letter to promote KC is found in the link below.
Parent letter II
by All Things Neonatal | May 7, 2017 | outcome, steroids
In our journey as Neonatologists and interdisciplinary teams we are forever seeking to rid or at least reduce the plague of BPD in the patients we care for. The PREMILOC trial was a double-blind, multicenter, randomized, placebo-controlled trial designed to test just that concept by introducing a low dose of hydrocortisone within 24 hours of birth. They enrolled infants born between 24 – 26+6 weeks of gestation and assigned them to receive either placebo or low-dose hydrocortisone 0.5mg/kg twice per day for 7 days, followed by 0.5 mg/kg per day for 3 days. The trial has been the subject of a previous post A Shocking Change in Position. Postnatal steroids for ALL microprems? Although the trial was stopped early due to financial concerns the authors demonstrated a 9% reduction in BPD using this strategy. The theory here in part is that the presence of hydrocortisone reduces inflammation and that this in turn may allow for better growth of lungs with time.
Why Not Adopt The Results Based on These Fantastic Results?
Steroids in preterm infants have a bad name. As discussed in previous posts on the topic the concern in all trials has been the potential impact of such medications on the developing brain. A nice summary of these concerns can be found in a paper in the CMAJ by the other “Canadian Neonatal Blogger” from 2001 in which he quoted the risk of cerebral palsy increasing from about 1 in 6 babies to 1 in 3 if babies born at < 28 weeks were exposed to postnatal steroids. Neurodevelopmental impairment overall would change from 1 in 4 to 1 in 3 if such exposure occurred. This paper and others expressing concerns over the effect of postnatal steroids led to a change in practice from more ubiquitous use to one restrained to only in those cases where the patient was nearing the end of all other options. This meant holding out for such therapy until such patients were at 90% or more O2 and on high mean airway pressures. Although not formally studied I was very concerned at the time with using this approach as I felt it was a “fait de complet” that they would either die or have significant developmental impairment should they survive due to the complications of having such severe BPD. It is critical to note though that the outcomes from these long term studies were in infants exposed to much longer courses of dexamethasone and at high doses that are used today.
Over the years with the development of the DART protocol and other more gentle approaches to steroids we as a group relaxed and certainly rescue courses of lower dose steroids have crept into practice when patients seem to be “stuck” on the ventilator.
Drumroll Please…
The results of the PREMILOC follow-up study are now here and in short they look good. Patients were followed up at an average age of 22 months and included a medical history, anthropometric measures, respiratory status, standardized neurological examination based on specific definitions of disabilities, and quantitative neurodevelopmental assessment using the revised Brunet-L.zine (RBL) scale. Follow-up was 93% in the hydrocortisone and 90% in the placebo arm which is important as we need not worry about the missed patients influencing the results to a significant degree if they had been included. Although some post-hoc analyses were done what I am most interested in is the primary outcome which is shown below.
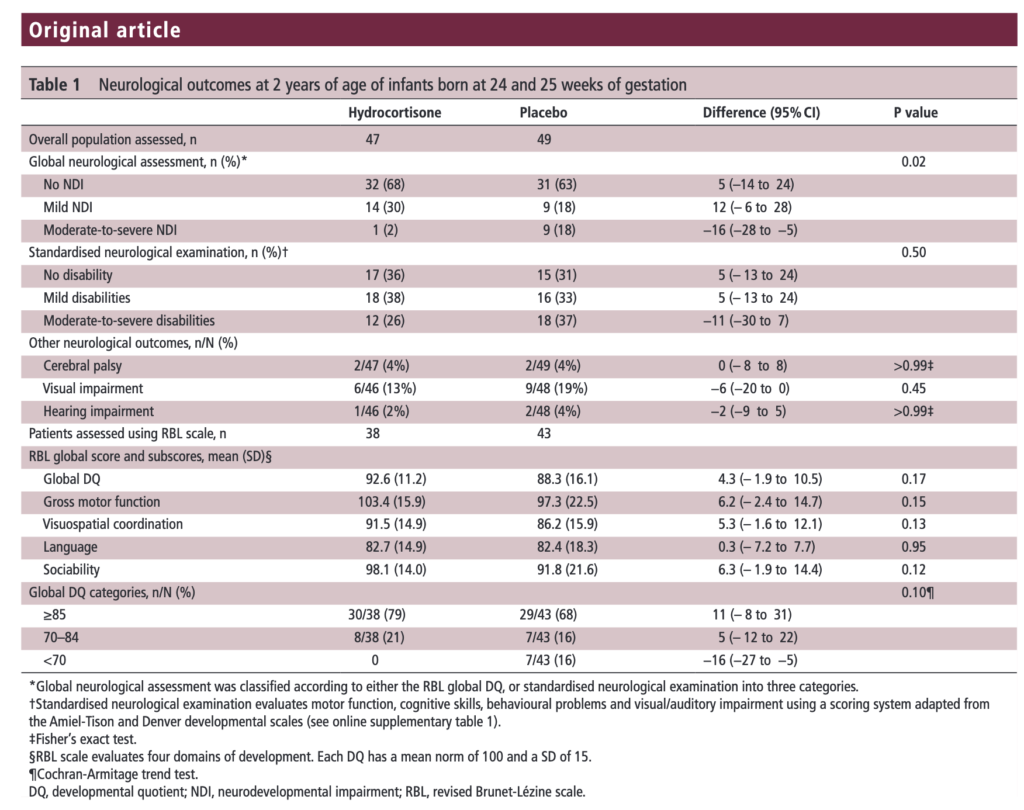
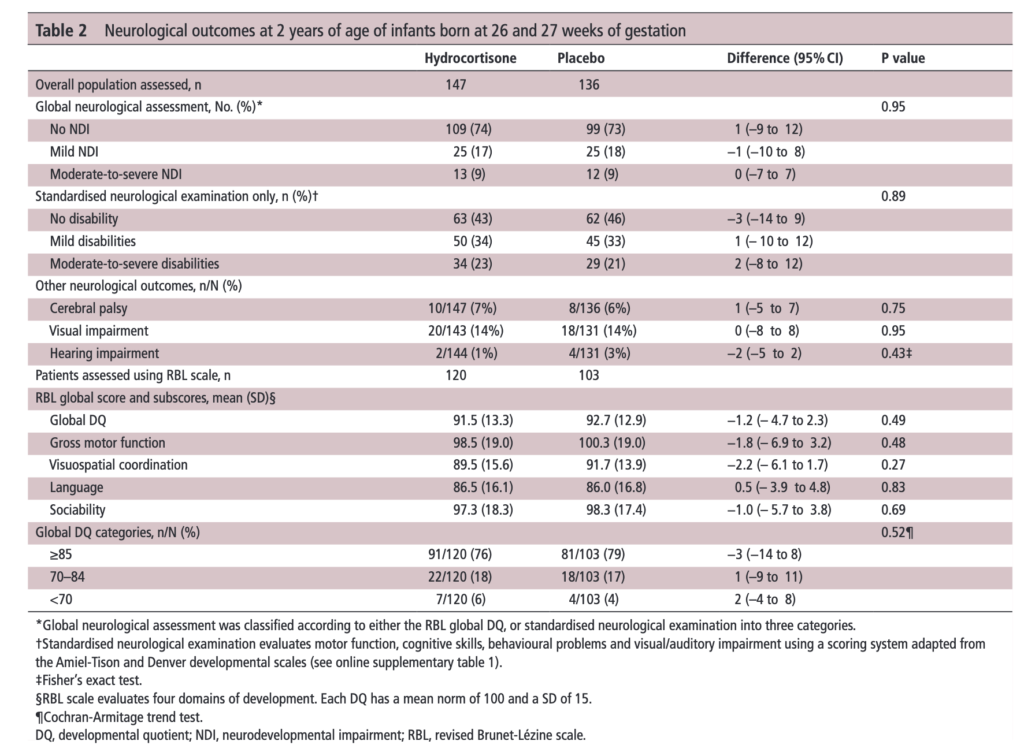 There was no difference in either neurodevelopment overall or any of the subcategories. This provides a great deal of reassurance to those who provide steroids this way. There will be those however that argue the study is too small. While a larger study might be better able to address whether there is a small difference in outcome I don’t think we will see one anytime soon. It is one of the challenges we face in Neonatology. Unlike the adult world with studies of thousands of patients, due to the small number of patients born at <28 weeks it is always a challenge to recruit into such large volume trials. We can compare trials by doing meta analyses or systematic reviews and perhaps that is where we will head with this study although given that different steroids will have been used (thinking dexamethasone as in the DART study) this will always be left open to question.
There was no difference in either neurodevelopment overall or any of the subcategories. This provides a great deal of reassurance to those who provide steroids this way. There will be those however that argue the study is too small. While a larger study might be better able to address whether there is a small difference in outcome I don’t think we will see one anytime soon. It is one of the challenges we face in Neonatology. Unlike the adult world with studies of thousands of patients, due to the small number of patients born at <28 weeks it is always a challenge to recruit into such large volume trials. We can compare trials by doing meta analyses or systematic reviews and perhaps that is where we will head with this study although given that different steroids will have been used (thinking dexamethasone as in the DART study) this will always be left open to question.
Is it worth it?
I suppose the real question here is the following for a parent to consider. “Would you like your baby to receive hydrocortisone shortly after birth with a 7% reduction in the risk of BPD at 36 weeks bearing in mind that although we don’t think there is an impact on long term development we aren’t certain yet”.
I guess to answer this question you need to think about the first part of the question. Is BPD at 36 weeks a good outcome to look at for benefit? The Canadian Neonatal Network has recently called for a rethink on this The New BPD That Matters. It turns out that it is 40 weeks and not 36 weeks that has the greatest prediction for respiratory morbidity after discharge. If you were to move the goal post to 40 weeks from 36 I strongly suspect one would see the 9% reduction in BPD as shown in the PREMILOC trial vanish. If that is the case, would a slightly earlier extubation time be enough to motivate families to take the plunge?
Although I often cringe at the expression “more trials are needed”, I think at least a combination of studies to achieve greater confidence in outcome may be needed. Barring that, we might just have to sit tight and accept that while there may be a little bit to be gained with the use of the PREMILOC protocol it may just not be enough to be clinically warranted at this time. May want to wait for the next big thing to tackle BPD…
by All Things Neonatal | Apr 12, 2017 | HIE, outcome, Uncategorized
The human body truly is a wondrous thing. Molecules made from one organ, tissue or cell can have far reaching effects as the products take their journey throughout the body. As a medical student I remember well the many lectures on the kidney. How one organ could control elimination of waste, regulate salt and water metabolism, blood pressure and RBC counts was truly thought provoking. At the turn of the century (last one and not 1999 – 2000) Medical school was about a year in length and as the pool of knowledge grew was expanded into the three or four year program that now exists. Where will we be in another 100 years as new findings add to the ever growing volume of data that we need to process? A good example of the hidden duties of a molecule is erythropoetin (Epo) the same one responsible from stimulating red blood cell production.
Double Duty Molecule
In saying that I am simplifying it as there are likely many processes this one hormone influences in the body but I would like to focus on its potential role in neuroprotection. In 1999 Bernaudin Et al performed an animal study in mice to test this hypothesis. In this elegant study, strokes were induced in mice and the amount of Epo and Epo receptors measured in injured tissues. Levels of both increased in the following way “endothelial cells (1 day), microglia/macrophage-like cells (3 days), and reactive astrocytes (7 days after occlusion)”. To test the hypothesis that the tissues were trying to protect themselves the authors then administered recombinant human Epo (rhEpo) to mice prior to inducing stroke and the injury was clearly reduced. This established Epo as a potential neuroprotectant. Other animal studies then followed demonstrating similar findings.
A Human Trial
When you think about hypoxic ischemic encephalopathy (HIE) you can’t help but think of whole body cooling. The evidence is pretty clear at this point that cooling in this setting reduces the combined outcome of death or neurodevelopmental disability at 18 months with a number needed to treat of 7. The risk reduction is about 25% compared to not those not cooled so in other words there is room to improve. Roughly 30-40% of infants who are cooled with moderate to severe HIE will still have this outome which leaves room for improvement. This was the motivation behind a trial called High-Dose Erythropoietin and Hypothermia for Hypoxic-Ischemic Encephalopathy: A Phase II Trial. This was a small trial comparing 50 patients (24 treated with rhEpo and cooling to 26 given placebo) who were treated with 1000 U of rEpo on days 1,2,3,5 and 7. Primary outcome was neurodevelopment at 12 months assessed by the Alberta Infant Motor Scale (AIMS)and Warner Initial Developmental Evaluation. A significant improvement in a subset of mobility on the latter was found and a significant difference in the AIMS overall. An additional finding giving support for a difference was that blinded reviews of MRI scans demonstrated a singificant improvement in brain tissue in those who received rhEPO. One curious finding in this study was that the mean timing of administration of rhEPO was 16.5 hours of life. Knowing that the benefit of cooling is best when done before 6 hours of age one can only wonder what impact earlier administration of a neuroprotective agent might have. This suggests that the addition of rEPO to cooling has additional impact but of course being a small study further research is needed to corroborate these findings.
The Next Step
This past week Malla et al published an interesting paper to add to the pool of knowledge in this area; Erythropoietin monotherapy in perinatal asphyxia with moderate to severe encephalopathy: a randomized placebo-controlled trial. This study was done from the perspective of asking if rhEPO by itself in resource poor settings without access to cooling in and of itself could make a difference in outcome for patients with HIE. This was a larger study with 100 Hundred term neonates (37 weeks or greater) with moderate or severe HIE. Fifty were randomized by random permuted block algorithm to receive either rhEPO 500 U kg− 1 per dose IV on alternate days for a total of five doses with the first dose given by 6 h of age (treatment group) or 2 ml of normal saline (50 neonates) similarly for a total of five doses (placebo group) in a double-blind study. The primary outcome was combined end point of death or moderate or severe disability at mean age of 19 months and the results of this and other important outcomes are shown below.
| Outcome |
Treatment |
Placebo |
p |
| Death/disability (mod/severe HIE) |
40% |
70% |
0.003 |
| Death/disability (mod HIE only) |
21% |
61% |
0.004 |
| Cerebral Palsy |
23% |
45% |
0.04 |
| MRI abnormalities |
40% |
60% |
0.04 |
| Seizures treatment at 19 months |
19% |
43% |
0.03 |
To say that these results are impressive is an understatement. The results are on par with those of cooling’s effect on reduction of injury and improvement in outcome. When looking at the primary outcome alone the result in dramatic when put in perspective of looking at number needed to treat which is 4! This is significant and I can’t help but wonder if the impact of this medication is at least in part related to starting the dosing within the same window of effectiveness of therapeutic hypothermia. Importantly there were no adverse effects noted in the study and given that rhEpo has been used to treat anemia of prematurity in many studies and not found to be associated with any significant side effects I would say this is a fairly safe therapy to use in this setting.
Next Steps
I find this puts us in a challenging position. The academic purists out there will call for larger and well designed studies to test the combination of rhEPO and cooling both initiated within 6 hours of age. While it takes years to get these results might we be missing an opportunity to enhance our outcomes with this combination that is right in front of us. The medication in question other than raising your RBC count has little if any side effects especially when given for such a short duration and by itself and possibly with cooling increases the rate of neuroprotection already. I don’t know about you but I at least will be bringing this forward as a question for my team. The fundamental question is “can we afford to wait?”
by All Things Neonatal | Feb 15, 2017 | outcome, Uncategorized
As a Neonatologist I doubt there are many topics discussed over coffee more than BPD. It is our metric by which we tend to judge our performance as a team and centre possibly more than any other. This shouldn’t be that surprising. The dawn of Neonatology was exemplified by the development of ventilators capable of allowing those with RDS to have a chance at survival.  As John F Kennedy discovered when his son Patrick was born at 34 weeks, without such technology available there just wasn’t much that one could do. As premature survival became more and more common and the gestational age at which this was possible younger and younger survivors began to emerge. These survivors had a condition with Northway described in 1967 as classical BPD. This fibrocystic disease which would cripple infants gave way with modern ventilation to the “new bpd”.
As John F Kennedy discovered when his son Patrick was born at 34 weeks, without such technology available there just wasn’t much that one could do. As premature survival became more and more common and the gestational age at which this was possible younger and younger survivors began to emerge. These survivors had a condition with Northway described in 1967 as classical BPD. This fibrocystic disease which would cripple infants gave way with modern ventilation to the “new bpd”.
The disease has changed to one where many factors such as oxygen and chorioamnionitis combine to cause arrest of alveolar development along with abnormal branching and thickening of the pulmonary vasculature to create insufficient air/blood interfaces +/- pulmonary hypertension. This new form is prevalent in units across the world and generally appears as hazy lungs minus the cystic change for the most part seen previously. Defining when to diagnose BPD has been a challenge. Is it oxygen at 28 days, 36 weeks PMA, x-ray compatible change or something else? The 2000 NIH workshop on this topic created a new approach to defining BPD which underwent validation towards predicting downstream pulmonary morbidity in follow-up in 2005. That was over a decade ago and the question is whether this remains relevant today.
Benchmarking
I don’t wish to make light of the need to track our rates of BPD but at times I have found myself asking “is this really important?” There are a number of reasons for saying this. A baby who comes off oxygen at 36 weeks and 1 day is classified as having BPD while the baby who comes off at 35 6/7 does not. Are they really that different? Is it BPD that is keeping our smallest babies in hospital these days? For the most part no. Even after they come off oxygen and other supports it is often the need to establish feeding or adequate weight prior to discharge that delays things these days. Given that many of our smallest infants also have apnea long past 36 weeks PMA we have all seen babies who are free of oxygen at 38 weeks who continue to have events that keep them in hospital. In short while we need to be careful to minimize lung injury and the consequences that may follow the same, does it matter if a baby comes off O2 at 36, 37 or 38 weeks if they aren’t being discharged due to apnea or feeding issues? It does matter for benchmarking purposes as one unit will use this marker to compare themselves against another in terms of performance. Is there something more though that we can hope to obtain?
When does BPD matter?
The real goal in preventing BPD or at least minimizing respiratory morbidity of any kind is to ensure that after discharge from the NICU we are sending out the healthiest babies we can into the community. Does a baby at 36 weeks and one day free of O2 and other support have a high risk of coming back to the hospital after discharge or might it be that those that are even older when they free of such treatments may be worse off after discharge. The longer it takes to come off support one would think, the more fragile you might be. This was the goal of an important study just published entitled Revisiting the Definition of Bronchopulmonary Dysplasia: Effect of Changing Panoply of Respiratory Support for Preterm Neonates. This work is yet another contribution to the pool of knowledge from the Canadian Neonatal Network. In short this was a retrospective cohort study of 1503 babies born at <29 weeks GA who were assessed at 18-21 months of age. The outcomes were serious respiratory morbidity defined as one of:
(1) 3 or more rehospitalizations after NICU discharge owing to respiratory problems (infectious or noninfectious);
(2) having a tracheostomy
(3) using respiratory monitoring or support devices at home such as an apnea monitor
or pulse oximeter
(4) being on home oxygen or continuous positive airway pressure at the time of assessment
While neurosensory impairment being one of:
(1) moderate to severe cerebral palsy (Gross Motor Function Classification System ≥3)
(2) severe developmental delay (Bayley Scales of Infant and Toddler
Development Third Edition [Bayley III] composite score <70 in either cognitive, language, or motor domains)
3) hearing aid or cochlear implant use
(4) bilateral severe visual impairment
What did they find?
The authors looked at 6 definitions of BPD and applied examined how predictive they were of these two outcomes. The combination of oxygen and/or respiratory support at 36 weeks PMA had the greatest capacity to predict this composite outcome. It was the secondary analysis though that peaked my interest. Once the authors identified the best predictor of adverse outcome they sought to examine the same combination of respiratory support and/oxygen at gestational ages from 34 -44 weeks PMA. The question here was whether the use of an arbitrary time point of 36 weeks is actually the best number to use when looking at these longer term outcomes. Great for benchmarking but is it great for predicting outcome?
It turns out the point in time with the greatest likelihood of predicting occurrence of serious respiratory morbidity is 40 weeks and not 36 weeks. Curiously, beyond 40 weeks it becomes less predictive. With respect to neurosensory impairment there is no real difference at any gestational age from 34-44 weeks PMA.
From the perspective of what we tell parents these results have some significance. If they are to be believed (and this is a very large sample) then the infant who remains on O2 at 37 weeks but is off by 38 or 39 weeks will likely fair better than the baby who remains on O2 or support at 40 weeks. It also means that the risk of neurosensory impairment is largely set in place if the infant born at < 29 weeks remains on O2 or support beyond 33 weeks. Should this surprise us? Maybe not. A baby who is on such support for over 5 weeks is sick and as a result the damage to the developing brain from O2 free radical damage and/or exposure to chorioamnionitis or sepsis is done.
It will be interesting to see how this study shapes the way we think about BPD. From a neurosensory standpoint striving to remove the need for support by 34 weeks may be a goal worth striving for. Failure to do so though may mean that we at least have some time to reduce the risk of serious respiratory morbidity after discharge.
Thank you to the CNN for putting out what I am sure will be a much discussed paper in the months to come.
by All Things Neonatal | Feb 8, 2017 | Neonatal, Neonatology, outcome, preemie, Prematurity
Positive pressure ventilation puts infants at risk of developing chronic lung disease (CLD). Chronic lung disease in turn has been linked many times over, as a risk for long term impacts on development. So if one could reduce the amount of positive pressure breaths administered to a neonate over the course of their hospital stay, that should reduce the risk of CLD and by extension developmental impairment. At least that is the theory. Around the start of my career in Neonatology one publication that carried a lot of weight in academic circles was the Randomized Trial of Permissive Hypercapnia in Preterm Infants which randomized 49 surfactant treated infants to either a low (35-45) or high (45-55) PCO2 target with the thought being that allowing for a higher pCO2 should mean that lower settings can be used. Lower settings on a ventilator would lead to less lung damage and therefore less CLD and in turn better outcomes. The study in question did show that the primary outcome was indeed different with almost a 75% reduction in days of ventilation and with that the era of permissive hypercapnia was born.
The Cochrane Weigh in
In 2001 a systematic review including this and another study concluded that there was insufficient evidence to support the strategy in terms of a benefit to death or chronic lung disease. Despite this lack of evidence and a recommendation from the Cochrane group that permissive hypercapnia be used only in the context of well designed trials the practice persisted and does so to this day in many places. A little lost in this discussion is that while the end point above was not different there may still be a benefit of shorter term ventilation.
A modern cohort
It would be unwise to ignore at this point that the babies of the late 90s are different that the ones in the current era. Surfactant and antenatal steroid use are much more prevalent now. Ventilation strategies have shifted to volume as opposed to pressure modes in many centres with a shift to early use of modalities such as high frequency ventilation to spare infants the potential harm of either baro or volutrauma. Back in 2015 the results of the PHELBI trial were reported Permissive hypercapnia in extremely low birthweight infants (PHELBI): a randomised controlled multicentre trial. This large trial of 359 patients randomized to a high or low target pCO2 again failed to show any difference in outcomes in terms of the big ones “death or BPD, mortality alone, ROP, or severe IVH”. What was interesting about this study was that they did not pick one unified target for pCO2 but rather set different targets as time went on reflecting that with time HCO3 rises so what matters more is maintaining a minimum pH rather than targeting a pCO2 alone which als0 reflects at least our own centre’s practice. There is a fly in the ointment here though and that is that the control group has a fault (at least in my eyes)
| Day of life |
Low Target |
High Target |
| 1-3 |
40-50 |
55-65 |
| 4-6 |
45-55 |
60-70 |
| 7-14 |
50-60 |
65-75 |
In the original studies of permissive hypercapnia the comparison was of a persistent attempt to keep normal pCO2 vs allowing the pCO2 to drift higher. Although I may get some argument on this point, what was done in this study was to compare two permissive hypercapnia ranges to each other. If it is generally accepted that a normal pCO2 is 35-45 mmHg then none of these ranges in the low target were that at all.
How did these babies do in the long run?
The two year follow-up for this study was published in the last month; Neurodevelopmental outcomes of extremely low birthweight infants randomised to different PCO2 targets: the PHELBI follow-up study. At the risk of sounding repetitive the results of Bayley III developmental testing found no benefit to developmental outcome. So what can we say? There is no difference between two strategies of permissive hypercapnia with one using a higher and the other a lower threshold for pCO2. It doesn’t however address the issue well of whether targeting a normal pCO2 is better or worse although the authors conclude that it is the short term outcomes of shorter number of days on ventilation that may matter the most.
The Truth is Out There
I want to believe that permissive hypercapnia makes a difference. I have been using the strategy for 15 or so years already and I would like to think it wasn’t poor strategy. I continue to think it makes sense but have to admit that the impact for the average baby is likely not what it once was. Except for the smallest of infants many babies these days born at 27 or more weeks of gestation due to the benefits of antenatal steroids, surfactant and modern ventilation techniques spend few hours to days on the ventilator. Meanwhile the number of factors such chorioamniotitis, early and late onset sepsis and genetic predisposition affect the risks for CLD to a great degree in the modern era. Not that they weren’t at play before but their influence in a period of more gentle ventilation may have a greater impact now. That so many factors contribute to the development of CLD the actual effect of permissive hypercapnia may in fact not be what it once was.
What is not disputed though is that the amount of time on a ventilator when needed is less when the strategy is used. Let us not discount the impact of that benefit as ask any parent if that outcome is of importance to them and you will have your answer.
So has permissive hypercapnia failed to deliver? The answer in terms of the long term outcomes that hospitals use to benchmark against one and other may be yes. The answer from the perspective of the baby and family and at least this Neonatologist is no.






 There was no difference in either neurodevelopment overall or any of the subcategories. This provides a great deal of reassurance to those who provide steroids this way. There will be those however that argue the study is too small. While a larger study might be better able to address whether there is a small difference in outcome I don’t think we will see one anytime soon. It is one of the challenges we face in Neonatology. Unlike the adult world with studies of thousands of patients, due to the small number of patients born at <28 weeks it is always a challenge to recruit into such large volume trials. We can compare trials by doing meta analyses or systematic reviews and perhaps that is where we will head with this study although given that different steroids will have been used (thinking dexamethasone as in the DART study) this will always be left open to question.
There was no difference in either neurodevelopment overall or any of the subcategories. This provides a great deal of reassurance to those who provide steroids this way. There will be those however that argue the study is too small. While a larger study might be better able to address whether there is a small difference in outcome I don’t think we will see one anytime soon. It is one of the challenges we face in Neonatology. Unlike the adult world with studies of thousands of patients, due to the small number of patients born at <28 weeks it is always a challenge to recruit into such large volume trials. We can compare trials by doing meta analyses or systematic reviews and perhaps that is where we will head with this study although given that different steroids will have been used (thinking dexamethasone as in the DART study) this will always be left open to question.

 As John F Kennedy discovered when his son Patrick was born at 34 weeks, without such technology available there just wasn’t much that one could do. As premature survival became more and more common and the gestational age at which this was possible younger and younger survivors began to emerge. These survivors had a condition with Northway described in 1967 as classical BPD. This fibrocystic disease which would cripple infants gave way with modern ventilation to the “new bpd”.
As John F Kennedy discovered when his son Patrick was born at 34 weeks, without such technology available there just wasn’t much that one could do. As premature survival became more and more common and the gestational age at which this was possible younger and younger survivors began to emerge. These survivors had a condition with Northway described in 1967 as classical BPD. This fibrocystic disease which would cripple infants gave way with modern ventilation to the “new bpd”.