I don’t think you can be a Neonatal blogger without writing about the patent ductus arteriosus from time to time. It’s been a little while so when something floats past my desk that I find interesting I share it with you. When that article is Canadian and written by someone I collaborate with on the Canadian Pediatric Society Fetus and Newborn Committee I am even more apt to do so. In the last few years the idea of letting nature take its course with respect to the PDA has been growing. The evidence is lacking that treatment for most infants in the first two weeks of life makes a difference to important pulmonary outcomes ie. BPD. There is a growing movement asking whether treatment at all really makes a difference to these infants or whether we should just be managing the medical complications of increased pulmonary bloodflow with diastolic steal from the kidneys and intestine. The alternative of course is to treat these infants most commonly with NSAIDs and hope that side effects such as renal impairment and spontaneous intestinal perforation don’t happen. Full disclosure, I was raised to find the PDA and if hemodynamically significant treat it, so that has been my general approach. I am open to suggestion though so without further adieu let’s talk about a recent Quebec study on the topic.
University of McGill in Montreal
Anchored by Dr. Altit with the lead author being De Carvalho Nunes the experience at this hospital was recently published as Natural evolution of the patent ductus arteriosus in the
extremely premature newborn and respiratory outcomes. The authors looked a specific population of infants born at <29 weeks gestational age (214 infants in total) and importantly had a reasonable number of small infants >26 weeks at birth (84) to see what happened to their PDAs in the long run. Many years ago the unit adopted a non-intervention policy with respect to treatment of PDAs and since 2015 were in a new hospital. This afforded them the opportunity to look retrospectively at a modern cohort of infants all cared for in the same environment from Feb 2015 – Sept 2019 and see what happened to respiratory morbidity over time. While this was retrospective and lacks a control group the concept here was that one could look at the rate of BPD over time and see if it was static, rising or falling and in turn you could also compare to the Canadian Neonatal Network (not done in this study) to see if your approach was leading to all sorts of morbidity.
What did they find?
The authors chose to standardize the definition of BPD:
Grade I (nasal flow cannula <1 L/min with FiO2 ≤ 70%; nasal cannula with flow of 1 to 3 L/min and FiO2 between 22 and 29%; CPAP, noninvasive positive pressure ventilation [NIPPV] or nasal cannula with flow >3 L/min with FiO2 of 21%),
Grade II (nasal flow cannula <1 L/min with FiO2 > 70%; nasal cannula with flow of 1 to 3 L/min and FiO2 ≥ 30%;
CPAP, NIPPV or nasal cannula with flow >3 L/min with FiO2 between 22
and 29%; invasive mechanical ventilation [IMV] with FiO2 of 21%)
Grade III
(NIMV, NIPPV or nasal cannula with flow >3 L/min with FiO2 ≥ 30%, IMV
with FiO2 > 21%).
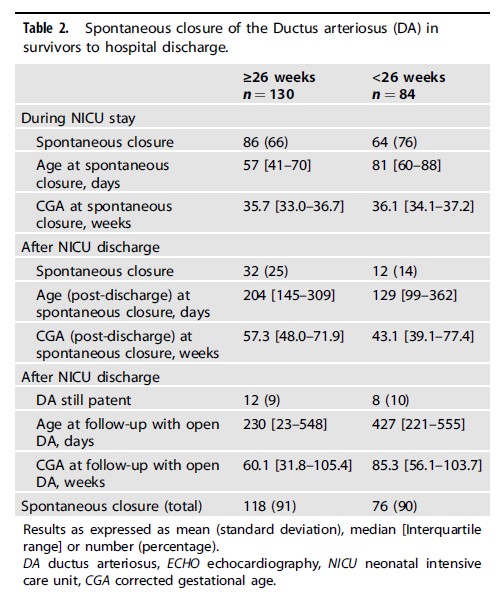
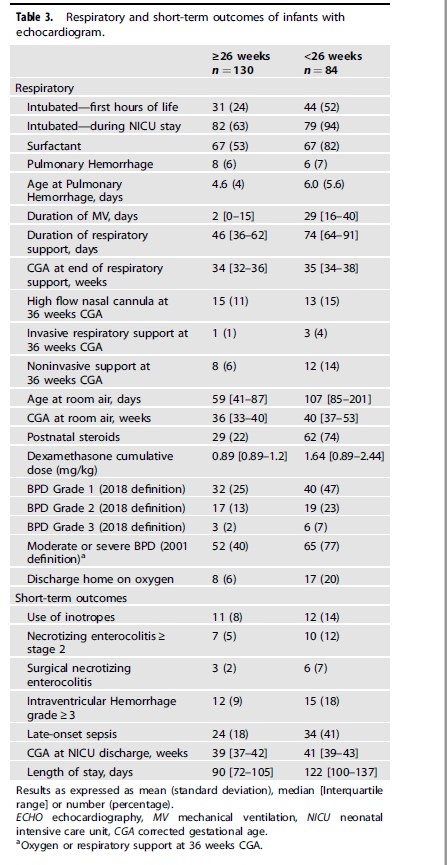
Looking at the respiratory outcomes a total of 77% had BPD under 26 weeks of varying severity compared to 40% in the larger infants. Other morbidities were not different.
Interestingly the authors also noted a decline in Grade 2 BPD over the 5 year study.
Thoughts on the results
It’s important to look at the overall results from the Canadian Neonatal Network to see how this group compares to the rest of the country. What follows is not perfect but its a start for a discussion.
One thing that I note though is that the rate of postnatal steroid use in this group was 75% under 26 weeks and 22% for those from 26-28 weeks. This represents a large increase over the mean in the CNN back in 2019 of 11.9% for postnatal steroid use. The babies under 26 weeks were also ventilated invasively for a median of 29 days. That seems a little long to me but there are no comparisons with the CNN to know for sure.
I can’t help but wonder if you are trading short term pain for long term gain. It’s hard to argue with the long term results in terms of a shift towards better rates of lower grade BPD. I do wonder though if the eventual closure of the PDA is being helped along with use of more postnatal dexamethasone. There is some data suggesting increased rates of closure with use of dexamethasone so maybe what is going on here is that rather than using NSAIDs there is a shift to long durations of ventilation and increased rates of dexamethasone use. Something for the authors to look at though.
With everything there are trade offs so maybe less NSAID use means longer ventilation and more postnatal steroids but in the end the pulmonary outcome is better? I see a prospective RCT coming to eventually settle this debate!


Is this not work that was done by the team at the McGill University Health Center in Montréal not Ste Justine?
good pick up! changed it