by All Things Neonatal | Mar 17, 2016 | antibiotics, MAS, Uncategorized
We live in a world at the moment where the public has become increasingly aware of the dangers of antibiotic overuse. Parents are more than ever requesting no erythromycin for the eyes after birth, and even on occasion questioning the need for antibiotics after delivery for the infant with risk factors for sepsis. The media has latched on to the debate as well by publishing the sensational articles about superbugs and medicine running out of the last lines of defence such as this article from the CBC.
As teams caring for newborns both preterm and term we are also increasingly aware of the dangers of altering the microbiome of these vulnerable infants with antibiotic overuse. Some babies robbed of the vaginal microbiome when delivery occurs by C-section, have their parents swabbing their newborn with vaginal secretions to populate their child with the “good bacteria” that come through a “natural” delivery although recent commentary questions the safety of such practice.
Infants born through meconium stained amniotic fluid can certainly become sick after delivery. Inhalation of meconium in the sickest infants often occurs during gasping episodes in utero after hypoxic stress causes evacuation of the rectal contents. The fetuses who inhale this material may go on to develop, inflammatory changes, areas of atelectasis and hyperinflation and pulmonary hypertension; the so called meconium aspiration syndrome. These infants of course may be extremely sick and need high frequency ventilation to manage their CO2 retention and in some cases may go on to ECMO although with inhaled nitric oxide this has become less common. As another consideration, could infection such as chorioamnionitis be the inciting event to cause passage of meconium in utero?
The health care team though for as long as I have been in practice would add to the treatment plan a course of antibiotics. In fact I would guess that many Neonatologists the world over have uttered the phrase “They are REALLY sick, please start antibiotics”. The real question though is whether the baby is in fact infected. Meconium is certainly a good growth medium for bacteria but with the short time from passage to delivery in most cases I doubt there is much time for significant growth. Moreover, I have found myself saying many times that such infants have a chemical pneumonitis and have often questioned whether antibiotics are really needed. Nonetheless it would take nerves of steel in some cases to not use antibiotics in these patients.
Then along came this study
Role of prophylactic antibiotics in neonates born through meconium-stained amniotic fluid (MSAF)—a randomized controlled trial by Goel A et al. This study was done prospectively by randomizing newborns born through meconium stained amniotic fluid to either antibiotics (N=121) for three days or no antibiotics (N=129) after diagnosis. In each case blood and CRP were drawn and if the infant was symptomatic (presence of respiratory distress, lethargy, abdominal distension, temperature or hemodynamic instability, hypoglycemia, apnea, or any other systemic abnormalities) a lumbar puncture and chest x-ray were added. The primary outcome variable was defined as ” the incidence of early (within first 72 h of birth) or late onset (after 72 h of birth) suspect sepsis (clinical symptoms or positive sepsis screen defined as ≥2 positive parameters) and confirmed sepsis (positive blood culture).”
Results
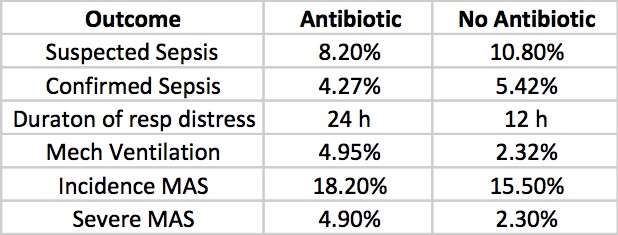
Clinicians in the study were allowed to continue antibiotics past the 72 hours or start antibiotics in the no antibiotic group if they considered an infant to have suspected sepsis or in fact were found to be proven. The outcomes for those possibilities are shown below.
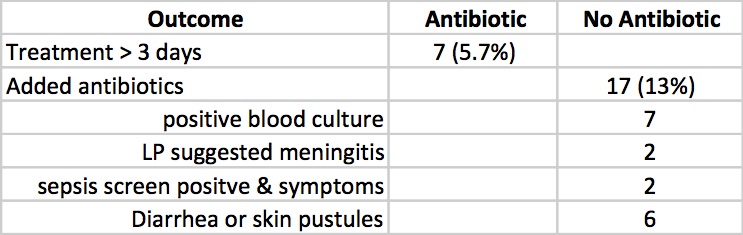
Taking it all together whether you started antibiotics or not the primary outcomes were no different. Furthermore there is no apparent harm based on outcomes that matter including the most important; death (3 in each group) that it does give one reason to pause when considering whether to treat prophylactically with antibiotics for babies born through meconium stained fluid.
What About The Sickest of The Sick
When attempting to answer this question the authors noted the following.
“On doing a subgroup analysis on incidence of sepsis in symptomatic babies (presenting with respiratory distress), both groups were found to have comparable incidence of suspect sepsis (p=0.084). The incidence of confirmed sepsis was more in symptomatic babies, although the total numbers was very few (p=0.01)”
Herein lies the challenge in declaring once and for all that we don’t need antibiotics at all in MAS. While the study was powered to adequately answer the primary outcome, the subgroups are so small that declaring with any confidence that one can stand by and watch infants with severe MAS without starting antibiotics is a tough conclusion to come to. The child though who is born through MSAF and has mild tachypnea as the only symptom I suspect is another story. I might even argue that the baby who is in need of CPAP could be watched and if they deteriorate have antibiotics started. As much as I would love to say none of these babies need antibiotics I would have to admit that I would cave once the baby was ventilated. It is better to provide a couple of days of antibiotics while awaiting blood cultures than to have a patient with sepsis left untreated or at least that is my opinion.
The question is what would you do?

by All Things Neonatal | Dec 29, 2015 | antibiotics, Neonatal
The scenario is often the same. Faced with a child born to a mother with risk factors for sepsis you decide to start antibiotics. The time comes closer to 36 – 48 hours when you must decide whether or not to continue. Each time we examine our results and look at cultures and try to do what is right. Yet defining right is sometimes hard for so many. If we had 100% sensitivity and specificity for all our tests it would be easy but we don’t. So what can we do?
If I had to have one wish though it would be that we could improve upon our diagnostic accuracy when it comes to treating suspected infections in the newborn. As health care providers we have an extremely loud inner voice trying to tell us to minimize risk when it comes to missing a true bacterial infection. On the other hand so much evidence has come forth in the last few years demonstrating that prolonging antibiotics beyond 48 hours is not just unwise in the absence of true infection but can be dangerous. Increased rates of necrotizing enterocolitis is just one such example but other concerns due to interfering with the newborn microbiome have arisen in more recent years. What follows are some general thoughts on septic workups that may help you (and myself in my own practice) as we move ahead into the New Year and may we cause less harm if we consider these.
The Role of Paired Blood Cultures
Although not published by our centre yet, we adopted this strategy for late onset sepsis a couple years back and have seen a significant reduction in work-ups deemed as true infections since adoption. While the temptation to do only one blood culture is strong as we have a desire to minimize skin breaks consider how many more there will be if you do one culture and get a CONS organism back. There will be several IV starts, perhaps a central line, repeat cultures etc. If you had done two at the start and one was positive and the other negative you could avoid the whole mess as it was a contaminant from the start. On my list of do no harms I think this may have the greatest benefit.
The Chest X-Ray Can Be Your Friend
While I am not a fan of routine chest x-rays I do believe that if you are prepared to diagnose an opacification on a chest x-ray as being due to a pneumonia (VAP or in those non-ventilated) that you need to follow this up with a repeat x-ray 24 – 48 hours later. 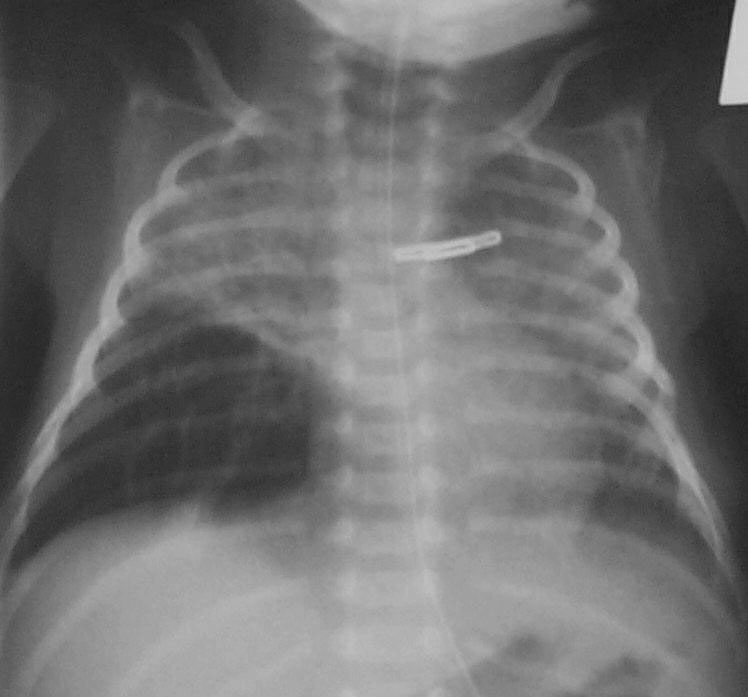 If the opacity is gone it was atelectasis as a true pneumonia will not clear that easily. Well worth the radiation exposure I say.
If the opacity is gone it was atelectasis as a true pneumonia will not clear that easily. Well worth the radiation exposure I say.
If You Are Going To Do a Work-up Make It A Complete One
We hear often in rounds the morning after a septic work-up that the baby was too sick to have an LP and that we can just check the CSF if the blood is positive. There are two significant problems to this approach. The first which is a significant concern is that in a recent study of patients with GBS meningitis, 20% of those who had GBS in the CSF had a negative blood culture. Think about that one clearly… relying on a positive culture to decide to continue antibiotics may lead to partially treated GBS meningitis when you discontinue the antibiotics prematurely. Not a good thing. The second issue is that infants with true meningitis can have relatively low CSF WBC counts and may drift lower with treatment. Garges et al in a review of 95 neonates with true meninigits found that CSF WBC counts >21 cells per mm3 had a sensitivity of 79% and specificity at 81%. This means that in those with true meningitis 19% of the time the WBC counts would be below 21 leading to the false impression that the CSF was “fine”. If antibiotics were effective it could well be by 48 hours that the negative CSF culture you find would incorrectly lead you to stop antibiotics. Message: Do the CSF sampling at the time of the septic work-up whenever possible.
If We Aren’t Prepared To Do a Supra Pubic Aspirate Should We Not Collect Urine At All?
This provocative question was asked by a colleague last week and is based on the results of a study which was the topic of the following post: Bladder Catherterizations for UTI: Causing more harm than good? The gist of it is that it would appear that in many cases the results of a catheter obtained urine cannot be trusted. If that is the case then are we ultimately treating infections that don’t actually exist when the only positive culture is from a urine. I believe using point of care ultrasound to obtain specimens from a SPA will be the way to go but in the meantime how do we address the question of whether a UTI is present or not? May need to rely on markers of inflammation such as a CRP or procalcitonin but that is not 100% sensitive or specific either but may be the best we have at the moment to determine how to interpret such situations.
Lastly, Slow Down And Practice Good Hand Hygiene
So much of what I said above is important when determining if an infection is present or not. The importance of preventing infection cannot be understated. Audits of hand hygiene practice more often than not demonstrate that physicians are a group with some of the lowest rates of compliance. Why is that? As a physician I think it has nothing to do with ignorance about how to properly perform the procedure but rather a tendency to rush from patient to patient in order to get all the things done that one needs to do well on service or call. If we all just slow down a little we may eventually have less need to run from patient to patient as the rate of infections may drop and with it demand for our time.
If slowing down is something that you too think is a good idea you may want to have a look at the book In Praise of Slowness by Carl Honore (TED Talk by Carl Below) which may offer some guidance how to do something that is more easily said than done. Here is hoping for a little slower pace in the new year. We could reap some fairly large benefits!
by All Things Neonatal | Oct 8, 2015 | antibiotics
The literature over the last few years has ballooned with reports of adverse outcomes related to antibiotic overuse. The most common adverse outcome noted on the worldwide stage is that of antimicrobial resistance and the so-called “superbugs” but in the NICU a whole host of other outcomes have been noted. Prolonging antibiotics beyond the first couple days of life has led to changes in the microbiome towards more pathogenic bacteria in the NICU. This shift has been accompanied by a rise in the rate of necrotizing enterocolitis in such units as well as many outcomes including some which affect us as adults such as asthma, which has been discussed on my Facebook page before.
New Consequences of Prolonging Antibiotics Beyond 48 hours
This month a colleague with a particular interest in this story forwarded our team the following paper. Prolonged Early Antibiotic Use and Bronchopulmonary Dysplasia in Very Low Birth Weight Infants by Novitsky et al. What this retrospective study demonstrated (comparing 747 infants treated for less than 48 hours to 159 treated beyond) was that the prolongation of antibiotics beyond the first 48 hours was associated with increased propensity to develop BPD. This finding remained true even when factoring in other risks for such outcomes including gestational age, maternal antibiotics, inborn status, clinical chorioamnionitis, prolonged rupture of membranes, preeclampsia, cesarean delivery, Apgar at 5 minutes, SNAP score, and mechanical ventilation. Curiously it is not just the initial course that is of concern but ongoing exposure as well. “After adding total cumulative hospital course antibiotic days, the odds of BPD remained increased with > 48 hours of antibiotic coverage in the 1st week. Furthermore, for every additional day of antibiotic coverage after the 1st week of life, there was an increased odds of developing BPD (OR, 1.16 per antibiotic day, 95% CI, 1.11–1.2). “
Prolonging antibiotics has another downstream consequence of increasing the rates of endotracheal tube colonization with bacteria to the tune of 38 vs. 16%. Additionally there was a shift in endotracheal tube culture patterns towards the emergence of resistant gram negative organisms (7% vs. 2%) if prolonged treatment up to 7 days was chosen. As has been shown in other studies rates of NEC were increased as well which should be no surprise based on previous similar work.
Wanting To Be Safe And Do The Right Thing
Once bitten twice shy as the saying goes. Any clinician who has missed an episode of sepsis in a newborn after not starting antibiotics is no doubt scarred to some degree. I have no doubt that all of us wish to do the right thing and protect the infants in our care when risk factors for sepsis indicate to us that there is a higher likelihood of sepsis being present. Looking at this study one can see the following risks were indeed more prevalent in the group treated beyond 48 hours.
| Risk |
Antibiotics < 48 hours in 1st week |
Antibiotics > 48 hours in 1st week |
P value |
| Birth weight |
1053 +/- 296 |
944 +/- 274 |
<0.01 |
| SNAP score |
13 +/- 9.2 |
17.8 +/-7.1 |
<0.01 |
| Clinical Chorio % |
5 |
18 |
<0.01 |
| Prolonged ROM |
14 |
26 |
<0.01 |
From the table I think we can agree that these infants were smaller and potentially sicker which likely motivated those clinicians to try and “do the right thing” even if cultures were negative. In this paper they defined sepsis as a positive culture and the presence of signs suggestive of sepsis, which avoids any confusion, related to such subjective classification as “possible or probable” sepsis where the clinical picture is suggestive but the culture negative. Despite a perceived increase in risk of sepsis after birth how accurate were clinicians? Early and Late Onset Sepsis Rates 14% in the < 48 hr and 16% in the > 48 hour group. Not significant at all.
The Cost of Fear
What is significant is the consequence of fear that motivates such decisions in the face of negative blood cultures. There is no difference in our ability to predict, yet doing so increases the risk of recovering bacteria from the endotracheal tube and more so resistant ones. The likelihood of obtaining bacterial growth in the endotracheal tube is more than doubled; NEC increased 2.5 times, gram-negative resistance tripled and the odds of developing BPD approximately doubled with an increasing tendency to this outcome simply by prolonging antibiotics. The cost of fear is that we trade our poor ability to truly predict sepsis with significant adverse risk that impacts the infant not only during their stay but long after they leave the NICU.
It is human nature to wish to do no harm. In order to truly achieve this we have to quell our need to “feel good” by knowing we have covered for sepsis and replace this with the fear of causing serious harm from such action. The decision to prolong the antibiotic course is the easy route as we go home comfortable that the baby is “covered” in case we are wrong. It makes us feel better but at significant cost. Only by changing our perspective of what constitutes harm will we ever move forward in this fight to change practice. Are you prepared to do it?





 If the opacity is gone it was atelectasis as a true pneumonia will not clear that easily. Well worth the radiation exposure I say.
If the opacity is gone it was atelectasis as a true pneumonia will not clear that easily. Well worth the radiation exposure I say.