In NICU we are always vigilant for infections. Bacterial sepsis is not uncommon and in fact in the latest 2020 Canadian Neonatal Network annual report, 9.4% of all babies across Canada born at < 33 weeks gestation experienced an infection after 3 days of age. Looking at the rate of infections in those with central lines (Central Line Bloodstream Infection or CLABSI the rate was 2.9/1000 line days. Infections in NICU are not surprising given that these infants often have need for vascular access and needle pokes breaking the skin and have a somewhat fragile immune system associated with premature birth. There are many strategies to reduce infection risk in the NICU but one in particular that has been around a long time is cleansing of the skin before any skin breaking procedures are performed such as for blood draws. Options exist for cleaning in terms of solution and whether it contains alcohol or not. A common skin antiseptic used is chlorhexidine which comes available as a 2% or 1% solution and with or without alcohol. The babies in particular who are our smallest may be sensitive to the alcohol and may be left with skin burns so for the smallest of infants we often clean without the alcohol containing solutions. What we are going to talk about today though is the use of 2% vs 1% and whether one is any different than the other in terms of effectiveness.
The Study
The study was done in India by Sharma A et al and entitled Aqueous chlorhexidine 1% versus 2% for neonatal skin antisepsis: a randomised non-inferiority trial. The authors set out to determine a number of things. The primary outcome was the percentage of negative skin swabs after application of both but they also looked for evidence of harm in terms of skin rashes and chlorhexidine blood levels. The strategy employed was for the investigator to identify a 4cm2 skin area to obtain the pre-antisepsis skin swab from one of the following sites: cubital fossa, dorsum hand or dorsum foot. Each patient could be enrolled again after a 96 hour period of time had elapsed. An alternate site was selected at the time of repeat enrolment. Each patient had a swab of this area done in a specific and repeated way in terms of strokes of the skin and then in a randomized fashion each patient received either the 1% or 2% solution on a swab. The solution was tested by a pharmacist throughout the study to validate the concentration of chlorhexidine (swab was dipped in the tested solution) and then applied in a consistent manner to the skin area. The area was left to dry for 60 seconds and then a second swab taken to determine whether the skin had been cleared of the bacteria that would have been picked up by the swab pre-chlorhexidine. The study set a target of a 5% non-inferiority limit comparing the two concentrations with an expected efficacy of about 90% for a 2% chlorhexidine solution to sterilize an area. This meant that if the efficacy of the 1% solution fell below a 5% difference it would be deemed to not be equivalent to the 2%.
What did they find?
First of all the groups were comparable in baseline characteristics. The babies enrolled in the study ranged from 26 0/7 to 42 6/7 weeks at delivery. As planned the groups; 341 to 1% and 344 to 2% were stratified for analysis into 26 0/7 to 27 6/7, 28 0/7 to 34 6/7 and 35 0/7 to 42 6/7 groups. The means GA and BW for each overall group however were 2018 vs 2029 and 34 vs 35 weeks for 1 and 2% groups.
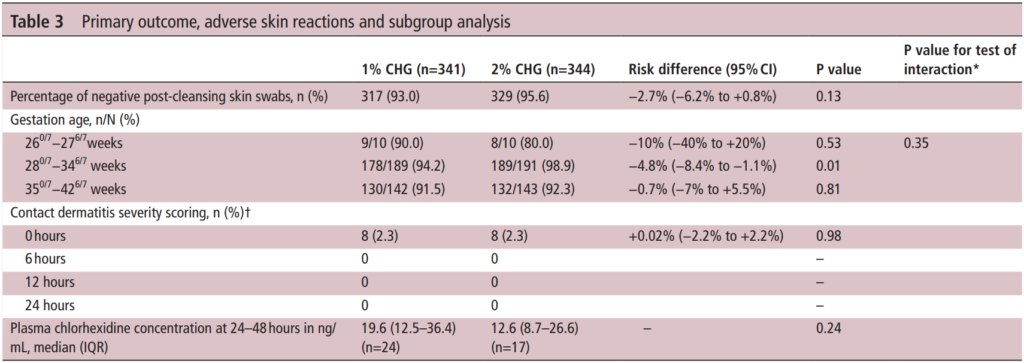
The table above has all the relevant information from the outcomes of interest. The efficacy of the antiseptics was a bit better than anticipated at 93% for the 1% vs 95.6% for the 2% group. On the surface you might be tempted to jump up and say “ah ha! They are equal”. However when you look at the 95% CI around the estimates you get a risk difference of -2.7% but the lower CI limit is -6.2% so as the authors concluded they are in fact not equal. Looking at the subgroups the number show some differences but only the middle group reached a statistical difference.
Importantly contact dermatitis which was scored by nursing using a standardized approach showed no difference at any time points. As expected some absorption did occur from application of the swabs but there was no difference between the two concentrations of significance although interestingly the higher concentration solution trended lower.
The authors do point out in this study that they did not get the 355 patients they wanted in each group although I have to say it was so close that I don’t believe adding a small percentage more of patients to the study would have been likely to change the conclusions.
Lesson from this study?
I suppose the first thing I take is that I am relieved that since our unit uses the 2% solution I don’t see any need for change. The second thing is that the rate of dermatitis from either solution is limited to about 1/10 babies after application but by 6 hours it is gone. Both have very high rates of successful skin antisepsis but if there is really no contraindication in terms of either skin irritation or high levels of drug in the blood my bias would be to grab onto the extra few percent successes at clearing the skin of bacteria. Even if it only a 3% gain, if we can maximize the cleaning of the skin before we poke a needle through it I think that is the way to go. If there are centres out there using the 1% though and plan on sticking to it I would be curious as to why?

