
by All Things Neonatal | Mar 14, 2018 | apnea of prematurity, caffeine, extubation, Neonatal, Neonatology, Prematurity, ventilation
Caffeine seems to be good for preterm infants. We know that it reduces the frequency of apnea in the this population and moreover facilitates weaning off the ventilator in a shorter time frame than if one never received it at all. The earlier you give it also seems to make a difference as shown in the Cochrane review on prophylactic caffeine. When given in such a fashion the chances of successful extubation increase. Less time on the ventilator not surprisingly leads to less chronic lung disease which is also a good thing.
I have written about caffeine more than once though so why is this post different? The question now seems to be how much caffeine is enough to get the best outcomes for our infants. Last month I wrote about the fact that as the half life of caffeine in the growing preterm infant shortens, our strategy in the NICU might be to change the dosing of caffeine as the patient ages. Some time ago though I wrote about the use of higher doses of caffeine and in the study analyzed warned that there had been a finding of increased cerebellar hemorrhage in the group randomized to receive the higher dosing. I don’t know about where you work but we are starting to see a trend towards using higher caffeine base dosing above 5 mg/kg/d. Essentially, we are at times “titrating to effect” with dosing being as high as 8-10 mg/kg/d of caffeine base.
Does it work to improve meaningful outcomes?
This month Vliegenthart R et al published a systematic review of all RCTs that compared a high vs low dosing strategy for caffeine in infants under 32 weeks at birth; High versus standard dose caffeine for apnoea: a systematic review. All told there were 6 studies that met the criteria for inclusion. Low dosing (all in caffeine base) was considered to be 5- 15 mg/kg with a maintenance dose of 2.5 mg/kg to 5 mg/kg. High dosing was a load of 5 mg/kg to 40 mg/kg with a maintenance of 2.5 mg/kg to 15 mg/kg. The variability in the dosing (some of which I would not consider high at all) makes the quality of the included studies questionable so a word of warning that the results may not truly be “high” vs “low” but rather “inconsistently high” vs. “inconsistently low”.
The results though may show some interesting findings that I think provide some reassurance that higher dosing can allow us to sleep at night.
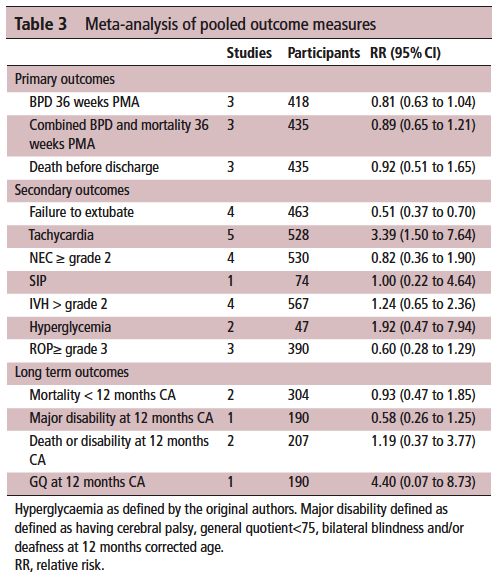
On the positive front, while there was no benefit to BPD and mortality at 36 weeks PMA they did find if they looked only at those babies who were treated with caffeine greater than 14 days there was a statistically significant difference in both reduction of BPD and decreased risk of BPD and mortality. This makes quite a bit of sense if you think about it for a moment. If we know that caffeine improves the chances of successful extubation and we also know it reduces apnea, then who might be on caffeine for less than 2 weeks? The most stable of babies I would expect! These babies were all < 32 weeks at birth. What the review suggests is that those babies who needed caffeine for longer durations benefit the most from the higher dose. I think I can buy that.
On the adverse event side, I suppose it shouldn’t surprise many that the risk of tachycardia was statistically increased with an RR of 3.4. Anyone who has explored higher dosing would certainly buy that as a side effect that we probably didn’t need an RCT to prove to us. Never mind that, have you ever taken your own pulse after a couple strong coffees in the morning?
What did it not show?
It’s what the study didn’t show that is almost equally interesting. The cerebellar hemorrhages seen in the study I previously wrote about were not seen at all in the other studies. There could be a lesson in there about taking too much stock in secondary outcomes in small studies…
Also of note, looking at longer term outcome measures there appears to be no evidence of harm when the patients are all pooled together. The total number of patients in all of these studies was 620 which for a neonatal systematic review is not bad. A larger RCT may be needed to conclusively tell us what to do with a high and low dosing strategy that we can all agree on. What do we do though in the here and now? More specifically, if you are on call tomorrow and a baby is on 5 mg/kg/d of caffeine already, will you intubate them if they are having copious apneic events or give them a higher dose of caffeine when CPAP or NIPPV that they are already on isn’t cutting it? That is where the truth about how you feel about the evidence really comes out. These decisions are never easy but unfortunately you sometimes have to make a decision and the perfect study hasn’t been done yet. I am not sure where you sit on this but I think this study while certainly flawed gives me some comfort that nothing is truly standing out especially given the fact that some of the “high dose” studies were truly high. Will see what happens with my next patient!
by All Things Neonatal | Jan 24, 2018 | caffeine, Neonatal, Neonatology, preemie, Prematurity
This has been a question that has befuddled Neonatologists for years. Get ten of us in a room and you will get a variety of responses ranging from (talking about caffeine base) 2.5 mg/kg/day to 10 mg/kg/day. We will espouse all of our reasons and question the issue of safety at higher doses but in the end do we really know? As I was speaking to a colleague in Calgary yesterday we talked about how convinced we are of our current management strategies but how we both recognize that half of what we think we know today we will be questioning in 10 years. So how convinced should we really be about caffeine?
Even the Cochrane Review Suggests There Is Something Amiss
Back in 2010 the Cochrane Collaboration examining 6 trials on caffeine for treating apnea of prematurity concluded “Methylxanthine is effective in reducing the number of apnoeic attacks and the use of mechanical ventilation in the two to seven days after starting treatment.” Notice the bolded section. Two to seven days. Interesting that we don’t see the effect last in perpetuity. Why might that be? Do babies become resistant with time or is there a change in the way these infants metabolize the drug such that levels in the bloodstream drop after that time point. It is almost certainly the latter and in the last 7 years have we really seen any response to this finding? I would say no for the most part although I don’t work in your unit so hard to say for sure. At least where I practice we pick a dose somewhere between 2.5-5 mg/kg/day and give a load of 10 mg/kg when we start the drug. From time to time we give a miniload of 5 mg/kg and may or may not increase the dose of maintenance based on the number of apneic events the babies are having. What if we could be proactive instead of reactive though. Do the babies need to have multiple events before we act or could we prevent the events from happening at all?
Proactive Treatment With Caffeine
We have known that caffeine clearance increases with postnatal age. The half-life of the drug shortens from about a week at the earliest gestational ages to 2-2.5 days by term equivalent age. For those infants who are older such as 32 weeks and above we expect them to be off caffeine (if they need it) within 2-3 weeks so I am not really talking about them but what about the babies born earlier than that or certainly MUCH earlier at 23 and 24 weeks who will be on caffeine possibly till term. Should one size (dose) fit all? No it really shouldn’t and some crafty researchers led by Koch G have published a paper that demonstrates why entitled Caffeine Citrate Dosing Adjustments to Assure Stable Caffeine Concentrations in Preterm Neonates.
In this paper the authors armed with knowledge of the half life of caffeine at different gestational ages were able to calculate the clearance of the drug at different postnatal ages to demonstrate in a model of a 28 week male infant weighing 1150g. The authors further took into account predicted weight changes and were able to calculate what the expected caffeine levels would be in the fictional infant at various time points. The target caffeine levels for this patient were a trough level of 15 -20 mg/L which are the currently acceptable ranges in the literature. The testing was first done using a standard load of 10 mg/kg (base) followed by 2.5 mg/kg/d (base) and demonstrated levels which yielded the following graph over time. 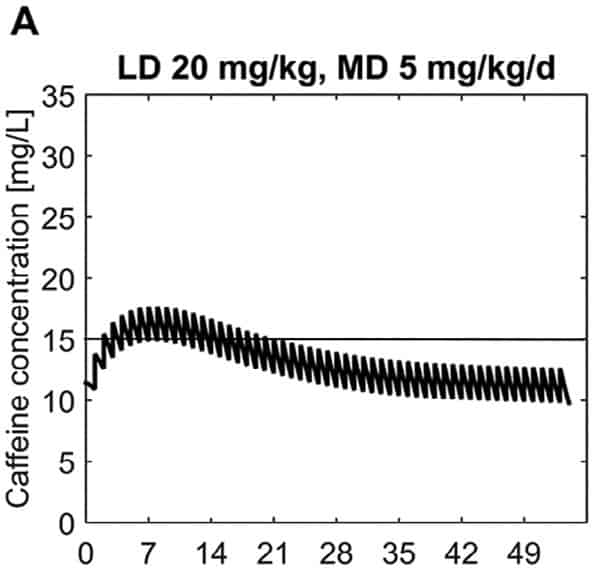 What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next.
What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next. 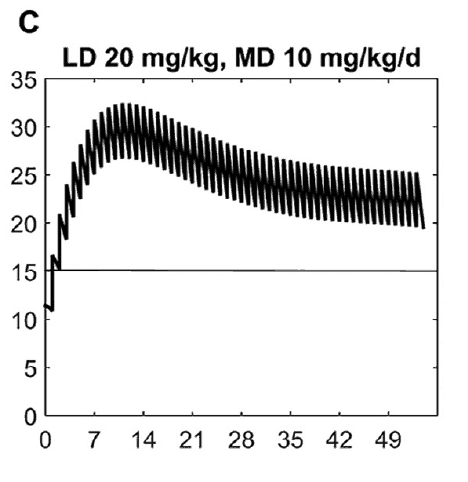 Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve.
Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve.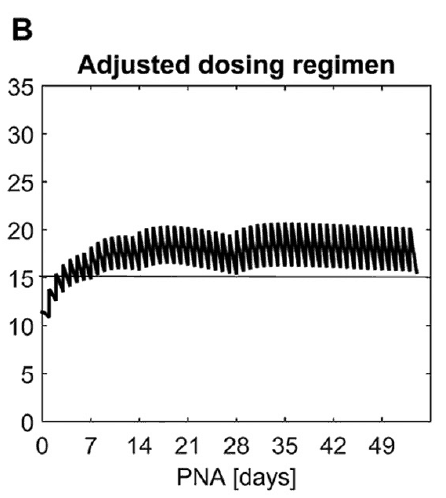 As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.
As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.
A Suggestion For The Future
What grabbed my attention here is the possibility that we could take a proactive rather than reactive approach to these infants. Once a small baby is controlled on their dose of caffeine whether it is 2.5, 3, 5 or even 6 mg/kg/d of caffeine should we wait for more events to occur and then react by increasing caffeine? What if we are too late to respond and the patient is intubated. What effect does this have on the developing lung, what about the brain that is subjected to bradycardic events with resultant drops in cardiac output and cerebral perfusion. Perhaps the solution is to work with our pharmacists and plan to increase dosing at several time points in the infants journey through the NICU even if they aren’t showing symptoms yet. No doubt this is a change in approach at least for the unit I work in but one that should start with a conversation!
by All Things Neonatal | Jan 15, 2018 | BPD, Neonatal, Neonatology, newborn, outcome, preemie, Prematurity, steroids
This must be one of my favourite topics as I have been following the story of early hydrocortisone to reduce BPD for quite some time. It becomes even more enticing when I have met the authors of the studies previously and can see how passionate they are about the possibilities. The PREMILOC study was covered on my site twice now, with the first post being A Shocking Change in Position. Postnatal steroids for ALL microprems? and the second reviewing the 22 month outcome afterwards /2017/05/07/early-hydrocortisone-short-term-gain-without-long-term-pain/.
The intervention here was that within 24 hours of birth babies born between 24-27 weeks gestational age were randomized to receive placebo or hydrocortisone 1 mg/kg/d divided q12h for one week followed by 0.5 mg/kg/d for three days. The primary outcome was rate of survival without BPD at 36 weeks PMA. The finding was a positive one with a 9% reduction in this outcome with the use of this strategy. Following these results were the two year follow-up which reported no evidence of harm but the planned analysis by gestational age groupings of 24-25 and 26-27 weeks was not reported at that time but it has just been released this month.
Is there a benefit?
Of the original cohort the authors are to be commended here as they were able to follow-up 93% of all infants studied at a mean age of 22 months. The methods of assessing their neurological status have been discussed previously but essentially comprised standardized questionnaires for parents, assessment tools and physical examinations.
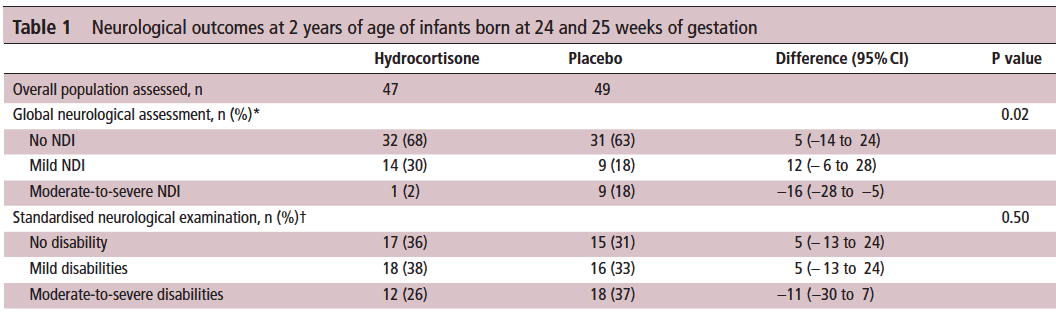
Let’s start off with what they didn’t find. There was no difference between those who received placebo vs hydrocortisone in the 26-27 week group but where it perhaps matters most there was. The infants born at 24-25 weeks are certainly some of our highest risk infants in the NICU. It is in this group that the use of hydrocortisone translated into a statistically significant reduction in the rate of neurodevelopmental impairment. The Global Neurological Assessement scores demonstrated a significant improvement in the hydrocortisone group with a p value of 0.02. Specifically moderate to severe disability was noted in 18% compared to 2% in the group receiving hydrocortisone.They did not find a difference in the neurological exam but that may reflect the lack of physical abnormalities with cognitive deficit remaining. It could also be explained perhaps by the physical examination not being sensitive enough to capture subtle differences.
Why might this be?
Adding an anti-inflammatory agent into the early phase of a preemies life might spare the brain from white matter damage. Inflammation is well known to inflict injury upon the developing brain and other organs (think BPD, ROP) so dampening these factors in the first ten days of life could bring about such results via a mechanism such as that. When you look at the original findings of the study though, a couple other factors also pop up that likely contribute to these findings as well. Infants in the hydrocortisone group had a statistical reduction in the rate of BPD and PDA ligations. Both of these outcomes have been independently linked to adverse neurodevelopmental outcome so it stands to reason that reducing each of these outcomes in the most vulnerable infants could have a benefit.
In fact when you add everything up, is there much reason not to try this approach? Ten days of hydrocortisone has now been shown to reduce BPD, decrease PDA ligations and importantly in the most vulnerable of our infants improve their developmental outcome. I think with this information at our fingertips it becomes increasingly difficult to ignore this approach. Do I think this will become adopted widely? I suspect there will be those who take the Cochrane approach to this and will ask for more well designed RCTs to be done in order to replicate these results or at least confirm a direction of effect which can then be studied as part of a systematic review. There will be those early adopters though who may well take this on. It will be interesting to see as these centres in turn report their before and after comparisons in the literature what the real world impact of this approach might be.
Stay tuned as I am sure this is not the last we will hear on this topic!

by All Things Neonatal | Jan 11, 2018 | NAS, Neonatal, neonatal abstinence, Neonatology, preemie
I wish it were otherwise, but in my practice, I have seen a growing number of pregnancies complicated by signs of substance withdrawal in newborn babies. Print, online, and broadcast news sources include regular reports on the “opioid crisis”. Data from the Canadian Institute for Health Information indicate that in 2016-17, about 1 in 200 newborns in Canada were affected by symptoms of drug withdrawal after birth. As this represents an average, there are no doubt some centres with much higher rates, while others may seem far lower depending on local usage patterns. Wherever you practice, if you care for newborns, you must learn how to treat this.
If you ask a physician in training how best to treat such conditions, their first response is often to use a medication such as morphine, thinking that it is best to treat an opioid withdrawal with the same class of drug. While this may be true, it is important to note that beginning with something much simpler, if not more natural, may reap tremendous benefits.
The Canadian Pediatric Society (CPS) released a new practice point this week, Managing infants born to mothers who have used opioids during pregnancy. While the document addresses the use of medical treatment, it highlights something far more important. Think of managing such pregnancies as a pyramid, with substance avoidance (the best strategy) on the bottom. The next level would be to manage newborns by keeping mothers and babies together. The top of the pyramid—that is, the fewest number of cases—would be treating these babies with medications.
For many families, avoidance is just not possible. Whether mothers use opioids due to addiction or chronic pain, it is simply unsafe to quit cold turkey. In October 2017, the Society of Obstetricians and Gynaecologists (SOGC) recommended against opioid detoxification in pregnancy because of the high risk of relapse. We should commend pregnant women who take responsibility for their health and seek care to stabilize on medications such as methadone or buprenorphine to manage their symptoms. After delivery, though, taking these babies and placing them on medications in a special care nursery should be a last resort.
Getting back to nature
Medications do work, but giving them means admitting babies to special care nurseries. This forced separation from families and, in particular, their mothers, actually leads to longer stays in hospital. Skin-to-skin care and breastfeeding contribute to better bonding between mother and child and have been associated with shortened hospital stays. In our centre, we have seen great success with many infants managed for up to seven days on the post-partum ward with their families. While this may seem like a long time, it is less than half of the average 15-day stay when babies are admitted to a special care unit.
Provided a mother is HIV-negative, the benefits of breastfeeding may go well beyond the bonding and closeness associated between mother and newborn. As most of these women continue to use a substance to ease their own withdrawal or pain, the small quantities of opioid that enter the breastmilk are in turn passed on to the newborn, which helps ease them through this transitional period in their life.
As the saying goes, sometimes less is more. In the case of caring for newborns exposed to opioids in pregnancy, getting back to nature and promoting skin-to-skin care and breastfeeding is just what this doctor ordered.
by All Things Neonatal | Dec 13, 2017 | BPD, Neonatal, Neonatology, preemie, ventilation
What is old is new again as the saying goes. I continue to hope that at some point in my lifetime a “cure” will be found for BPD and is likely to centre around preventing the disease from occurring. Will it be the artificial placenta that will allow this feat to be accomplished or something else? Until that day we unfortunately are stuck with having to treat the condition once it is developing and hope that we can minimize the damage. When one thinks of treating BPD we typically think of postnatal steroids. Although the risk of adverse neurodevelopmental outcome is reduced with more modern approaches to use, such as with the DART protocol,most practitioners would prefer to avoid using them at all if possible. We know from previous research that a significant contributor to the development of BPD is inflammation. As science advanced, the specific culprits for this inflammatory cascade were identified and leukotrienes in particular were identified in tracheal lavage fluid from infants with severe lung disease. The question then arises as to whether or not one could ameliorate the risk of severe lung disease by halting at least a component of the inflammatory cascade leading to lung damage.
Leukotriene Antagonists
In our unit, we have tried using the drug monteleukast, an inhibitor of leukotrienes in several patients. With a small sample it is difficult to determine exactly whether this has had the desired effect but in general has been utilized when “all hope is lost”. The patient has severe disease already and is stuck on high frequency ventilation and may have already had a trial of postnatal steroids. It really is surprising that with the identification of leukotriene involvement over twenty years ago it took a team in 2014 to publish the only clinical paper on this topic. A German team published Leukotriene receptor blockade as a life-saving treatment in severe bronchopulmonary dysplasia.in 2014 and to date as far as I can see remains the only paper using this strategy. Given that we are all looking for ways to reduce BPD and this is the only such paper out there I thought you might want to see what they found. Would this be worth trying in your own unit? Well, read on and see what you think!
Who was included?
This study had an unusual design that will no doubt make statistical purists cringe but here is what they did. The target population for the intervention were patients with “life threatening BPD”. That is, in the opinion of the attending Neonatologist the patient had a greater than 50% likelihood of dying and also had to meet the following criteria; born at < 32 weeks GA, <1500g and had to be ventilated at 28 days. The authors sought a blinded RCT design but the Research Ethics Board refused due to the risk of the drug being low and the patients having such a high likelihood of death. The argument in essence was if the patients were likely to die and this drug might benefit them it was unethical to deny them the drug. The authors attempted to enroll all eligible patients but wound up with 11 treated and 11 controls. The controls were patients either with a contraindication to the drug or were parents who consented to be included in the study as controls but didn’t want the drug. Therapy was started for all between 28 – 45 days of age and continued for a wide range of durations (111+/-53 days in the study group). Lastly, the authors derived a score of illness severity that was used empirically:
PSC = FiO2 X support + medications
– support was equal to 2.5 for a ventilator. 1.5 for CPAP and 1 for nasal cannulae or an oxygen hood
– medications were equal to 0.2 for steroids, 0.1 for diruetics or inhaled steroids, 0.05 for methylxanthines or intermittent diruetics.
Did it make a difference?
The study was very small and each patient who received the medication was matched with one that did not receive treatment. Matching was based on GA, BW and the PSC with matching done less than 48 hours after enrollment in an attempt to match the severity of illness most importantly.

First off survival in the groups were notably different. A marked improvement in outcome was noted in the two groups. Of the deaths in the control group, the causes were all pulmonary and cardiac failure, although three patients died with a diagnosis of systemic inflammatory response syndrome. That is quite interesting given that monteleukast is an anti-inflammatory medication and none of the patients in the treatment arm experienced this diagnosis.
The second point of interest is the trend in the illness severity score over time.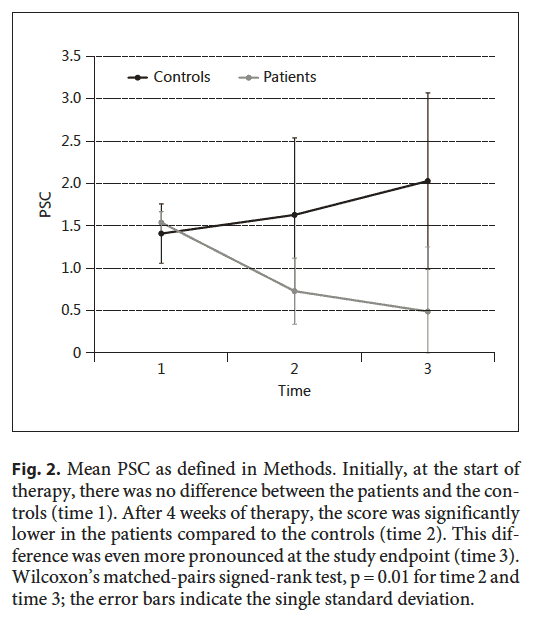 The time points in the figure are time 1 (start of study), time 2 (4 weeks of treatment), time 3 (end of treatment). These patients improved much more over time than the ones who did not receive treatment.
The time points in the figure are time 1 (start of study), time 2 (4 weeks of treatment), time 3 (end of treatment). These patients improved much more over time than the ones who did not receive treatment.
The Grain of Salt
As exciting as the results are, we need to acknowledge a couple things. The study is small and with that the risk of the results appearing to be real but in actual fact there being no effect is not minimal. As the authors knew who was receiving monteleukast it is possible that they treated the kids differently in the unit. If you believed that the medication would work or moreover wanted it to work, did you pay more attention on rounds and during a 24 hour period to those infants? Did the babies get more blood gases and tighter control of ventilation with less damage to the lungs over time? There are many reasons why these patients could have been different including earlier attempts to extubate. The fact is though the PSC scores do show that the babies indeed improved more over time so I wouldn’t write it off entirely that they did in fact benefit. The diagnosis of SIRS is a tough one to make in a newborn and I worry a little that knowing the babies didn’t receive an anti-inflammatory drug they were “given” that diagnosis.
Would I use it in spite of these faults? Yes. We have used it in such cases but I can’t say for sure that it has worked. If it does, the effect is not immediate and we are left once we start it not knowing how long to treat. As the authors here say though, the therapeutic risk is low with a possibly large benefit. I doubt it is harmful so the question we are left asking is whether it is right for you to try in your unit? As always perhaps a larger study will be done to look at this again with a blinded RCT structure as the believers won’t show up I suspect without one!
by All Things Neonatal | Nov 16, 2017 | Neonatal, Neonatology, preemie, Prematurity
Given that today is world prematurity day it seems fitting to talk about prematurity at the absolute extreme of it.
It has been some time since as a regional program we came to accept that we would offer resuscitation to preterm infants born as early as 23 weeks gestational age. This is perhaps a little later in the game that other centers but it took time to digest the idea that the rate of intact survival was high enough to warrant a trial of resuscitation. This of course is not a unilateral decision but rather a decision arrived at after consultation with the family and interprofessional team. To be sure it is not an easy one. Other centers have argued that resuscitation should be offered to those infants as young as 22 weeks gestational age and data now exists due to enough centres doing so to provide families with some guidance as to expected survival rates and importantly the likelihood of disability. This topic has been covered previously in /2015/09/25/winnipeg-hospital-about-to-start-resuscitating-infants-at-23-weeks/. Why cover this topic again? Well an article on CNN might have something to do with it.
Resuscitating Below 22 weeks
This week as I was perusing the news I came across a rather shocking article on CNN. Born before 22 weeks, ‘most premature’ baby is now thriving.  The article tells the tale of a baby delivered at 21 weeks and 4 days that now as a three year old is reaching appropriate milestones without any significant impairments. It is a story that is filled with inspiration and so I am not mistaken I am delighted for this child and their family that this outcome has occurred. When the lay press latches onto stories like this there is no doubt a great deal of sensationalism to them and in turn that gathers a lot of attention. This in turn is a great thing for media.
The article tells the tale of a baby delivered at 21 weeks and 4 days that now as a three year old is reaching appropriate milestones without any significant impairments. It is a story that is filled with inspiration and so I am not mistaken I am delighted for this child and their family that this outcome has occurred. When the lay press latches onto stories like this there is no doubt a great deal of sensationalism to them and in turn that gathers a lot of attention. This in turn is a great thing for media.
A Few Caveats Though
With the exception of pregnancies conceived through IVF the best dating we have is only good to about +/- 5 days when an early first trimester ultrasound is performed or the date of the last menstrual period is fairly certain. A baby though who is born at 21 weeks + 4 days may in fact be 22 +3 days or even more depending on when the dating was done (second trimester worse). Let’s not take away though from the outcome being this good even at 22 weeks. That is a pretty perfect outcome for this family but the point is that this baby may in fact be older than 21 weeks.
Secondly, there are millions of babies born each year in North America. Some of these infants are born at 22 weeks. How do they fare overall? From the paper by Rysavy et al from 2015 the results are as follows.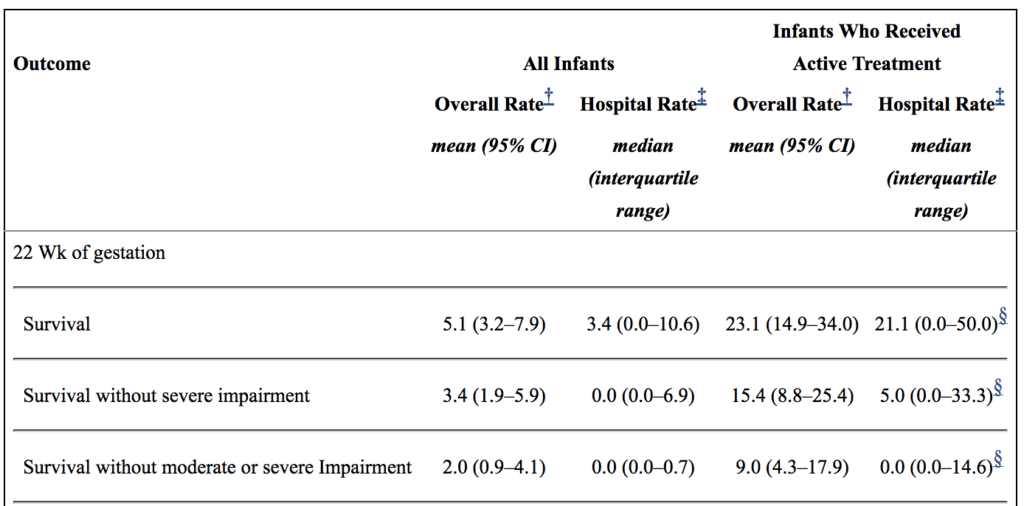
If you look at the overall rate of survival it is on an average of 5.1%. If you take a look though at those infants in whom resuscitation is provided that number increases to a mean of 23%. Intact survival is 9% overall. The odds aren’t great but they are there and I suspect the infant in the article is one of those babies. Flipping the argument though to the glass is half empty, 91% of infants born at 22 weeks by best estimate who are offered resuscitation will have a moderate or severe disability or die. I am not saying what one should do in this situation but depending on how a family processes the data they will either see the 110 chance of intact survival as a good thing or a 9/10 chance of death or disability as a very bad thing. What a family chooses though is anyone’s best guess.
Should we resuscitate below 22 weeks if the family wishes?
I guess in the end this really depends on a couple things. First off, how certain are the dates? If there is any degree of uncertainty then perhaps the answer is yes. If the dates are firm then I at least believe there is a barrier at which futility is reached. Perhaps this isn’t at 21 weeks as some patients may indeed be older but think about what you would offer if a family presented at 20 weeks and wanted everything done. What if it were 19 weeks? I suspect the point of futility for all lies somewhere between 19-21 weeks.
As I prepare to attend the annual meeting in Ottawa tomorrow for the Fetus and Newborn Committee I think it is prudent to point out just how difficult all of this is. The current statement on Counselling and management for anticipated extremely preterm birth I think hits on many of these issues. The statement is the product on not only the think tank that exists on this committee but was the product of a national consultation. I know I may be biased since I sit on the committee but I do believe it really hits the mark.
Should we be thinking about resuscitating at 21 weeks? For me the answer is one clouded by a whole host of variables and not one that can be easily answered here. What I do think though is that the answer in the future may be a yes provided such infants can be put onto an artificial placenta.  Even getting a few more weeks of growth before aerating those lungs is necessary may make all the difference. The NICUs of tomorrow certainly may look quite different than they do now.
Even getting a few more weeks of growth before aerating those lungs is necessary may make all the difference. The NICUs of tomorrow certainly may look quite different than they do now.




 What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next.
What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next.  Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve.
Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve. As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.
As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.




 The time points in the figure are time 1 (start of study), time 2 (4 weeks of treatment), time 3 (end of treatment). These patients improved much more over time than the ones who did not receive treatment.
The time points in the figure are time 1 (start of study), time 2 (4 weeks of treatment), time 3 (end of treatment). These patients improved much more over time than the ones who did not receive treatment.
 The article tells the tale of a baby delivered at 21 weeks and 4 days that now as a three year old is reaching appropriate milestones without any significant impairments. It is a story that is filled with inspiration and so I am not mistaken I am delighted for this child and their family that this outcome has occurred. When the lay press latches onto stories like this there is no doubt a great deal of sensationalism to them and in turn that gathers a lot of attention. This in turn is a great thing for media.
The article tells the tale of a baby delivered at 21 weeks and 4 days that now as a three year old is reaching appropriate milestones without any significant impairments. It is a story that is filled with inspiration and so I am not mistaken I am delighted for this child and their family that this outcome has occurred. When the lay press latches onto stories like this there is no doubt a great deal of sensationalism to them and in turn that gathers a lot of attention. This in turn is a great thing for media.
 Even getting a few more weeks of growth before aerating those lungs is necessary may make all the difference. The NICUs of tomorrow certainly may look quite different than they do now.
Even getting a few more weeks of growth before aerating those lungs is necessary may make all the difference. The NICUs of tomorrow certainly may look quite different than they do now.