
by All Things Neonatal | Apr 2, 2018 | congenital heart defects, Neonatal, Neonatology
In 2017 the Canadian Pediatric Society published the practice point Pulse oximetry screening in newborns to enhance detection of critical congenital heart disease. In this document we recommended universal screening for CCHDs but stressed the following:
“Recognizing that delivery and time of discharge practices vary across Canada, the timing of testing should be individualized for each centre and (ideally) occur after 24 hours postbirth to lower FP results. And because the intent is to screen newborns before they develop symptoms, the goal should be to perform screening before they reach 36 hours of age.”
This recommendation was put in place to minimize the number of false positive results and prevent Pediatricians and Cardiologists nationwide from being inundated with requests to rule out CCHD as earlier testing may pick up other causes for low oxygen saturation such as TTN. The issue remains though that many patients are indeed discharged before 24 hours and in the case of midwife deliveries either in centres or in the home what do we do?
A Population Study From the Netherlands May Be of Help Here
Researchers in the Netherlands had a golden opportunity to answer this question as a significant proportion of births occur there in the home under the care of a midwife. Accuracy of Pulse Oximetry Screening for Critical Congenital Heart Defects after Home Birth and Early Postnatal Discharge by Ilona C. Narayen et al was published this month in J Peds. About 30% of births are cared for by a midwife with about 20% occurring in the home. The authors chose to study this population of infants who were all above 35 weeks gestation and not admitted to an intensive care nor had suspicion of CCHD prior to delivery. The timing of the screening was altered from the typical 24-48 hours to be two time points to be more reflective of midwives practice. All patients were recruited after birth with the use of information pamphlets. The prospective protocol was screening on 2 separate moments: on day 1, at least 1 hour after birth, and on day 2 or 3 of life. The criteria for passing or failing the test are the same as those outlined in the CPS practice point. As part of the study, patients with known CCHDs were also screened separately as a different group to determine the accuracy of the screening test in patients with known CCHD.
Results
There were nearly 24000 patients born during this period. Only 49 cases of CCHD were identified by screening and of these 36 had been picked up antenatally giving a detection rate of 73%. Out of 10 patients without prenatal diagnosis who also had saturation results available the detection rate was 50%. Three of the misses were coarctation of the aorta (most likely diagnosis to be missed in other studies), pulmonary stenosis (this one surprises me) and TGA (really surprises me). The false-positive rate of pulse oximetry screening (no CCHD) was 0.92%. The specificity was over 99% meaning that if you didn’t have CCHD you were very likely to have a negative test. Not surprisingly, most false- positives occurred on day 1 (190 on day 1 vs 31 infants on day 2 or 3). There were five patients missed who were not detected either by antenatal ultrasound. These 5 negatives ultimately presented with symptoms at later time points and all but one survived (TGA) so out of 24000 births the system for detecting CCHD did reasonably well in enhancing detection as they picked up another 5 babies that had been missed antenatally narrowing the undetected from 10 down to 5.
Perhaps the most interesting thing about the study though is what they also found. As the authors state: “Importantly, 61% (134/221) of the infants with false-positive screenings proved to have significant noncardiac illnesses re- quiring intervention and medical follow-up, including infection/ sepsis (n = 31) and PPHN or transient tachypnea of the newborn (n = 88)”
There are certainly detractors of this screening approach but remember these infants were all thought to be asymptomatic. By implementing the screening program there was opportunity to potentially address infants care needs before they went on to develop more significant illness. Under appreciated TTN could lead to hypoxia and worsen and PPHN could become significantly worse as well. I think it is time to think of screening in this way as being more general and not just about finding CCHD. It is a means to identify children with CCHD OR RESPIRATORY illnesses earlier in their course and do something about it!

by All Things Neonatal | Apr 1, 2018 | Neonatal, Neonatology, preemie, Prematurity, ventilation
For almost a decade now confirmation of intubation is to be done using detection of exhaled CO2. The 7th Edition of NRP has the following to say about confirmation of ETT placement “The primary methods of confirming endotracheal tube placement within the trachea are detecting exhaled CO2 and a rapidly rising heart rate.” They further acknowledge that there are two options for determining the presence of CO2 “There are 2 types of CO2 detectors available. Colorimetric devices change color in the presence of CO2. These are the most commonly used devices in the delivery room. Capnographs are electronic monitors that display the CO2 concentration with each breath.” The NRP program stops short of recommending one versus the other. I don’t have access to the costs of the colorimetric detectors but I would imagine they are MUCH cheaper than the equipment and sensors required to perform capnography using the NM3 monitor as an example. The real question though is if capnography is truly better and might change practice and create a safer resuscitation, is it the way to go?
Fast but not fast enough?
So we have a direct comparison to look at. Hunt KA st al published Detection of exhaled carbon dioxide following intubation during resuscitation at delivery this month. They started from the standpoint of knowing from the manufacturer of the Pedicap that it takes a partial pressure of CO2 of 4 mm Hg to begin seeing a colour change from purple to yellow but only when the CO2 reaches 15 mm Hg do you see a consistent colour change with that device. The capnograph from the NM3 monitor on the other hand is quantitative so is able to accurately display when those two thresholds are reached. This allowed the group to compare how long it took to see the first colour change compared to any detection of CO2 and then at the 4 and 15 mm Hg levels to see which is the quicker method of detection. It is an interesting question as what would happen if you were in a resuscitation and the person intubates and swears that they are in but there is no colour change for 5, 10 or 15 seconds or longer? At what point do you pull the ETT? Compare that with a quantitative method in which there is CO2 present but it is lower than 4. Would you leave the tube in and use more pressure (either PIP/PEEP or both?)? Before looking at the results, it will not shock you that ANY CO2 should be detected faster than two thresholds but does it make a difference to your resuscitation?
The Head to Head Comparison
The study was done retrospectively for 64 infants with a confirmed intubation using the NM3 monitor and capnography. Notably the centre did not use a colorimetric detector as a comparison group but rather relied on the manufacturers data indicating the 4 and 15 mm Hg thresholds for colour changes. The mean age of patients intubated was 27 weeks with a range of 23 – 34 weeks. The results I believe show something quite interesting and informative.
|
Median time secs (range) |
| Earliest CO2 detection |
3.7 (0 – 44s) |
| 4 mm Hg |
5.3 (0 – 727) |
| 15 mm Hg |
8.1 (0 – 727) |
I wouldn’t worry too much about a difference of 1.6 seconds to start getting a colour change but it is the range that has me a little worried. The vast majority of the patients demonstrated a level of 4 or 15 mm Hg within 50 seconds although many were found to take 25-50 seconds. When compared to a highest level of 44 seconds in the first detection of CO2 group it leads one to scratch their head. How many times have you been in a resuscitation and with no CO2 change you keep the ETT in past 25 seconds? Looking closer at the patients, there were 12 patients that took more than 30 seconds to reach a threshold of 4 mm Hg. All but one of the patients had a heart rate in between 60-85. Additionally there was an inverse relationship found between gestational age and time to detection. In other words, the smallest of the babies in the study took the longest to establish the threshold of 4 and 15 mm Hg.
Putting it into context?
What this study tells me is that the most fragile of infants may take the longest time to register a colour change using the colorimetric devices. It may well be that these infants take longer to open up their pulmonary vasculature and deliver CO2 to the alveoli. As well these same infants may take longer to open the lung and exhale the CO2. I suppose I worry that when a resuscitation is not going well and an infant at 25 weeks is bradycardic and being given PPV through an ETT without colour change, are they really not intubated? In our own centre we use capnometry in these infants (looks for a wave form of CO2) which may be the best option if you are looking to avoid purchasing equipment for quantitative CO2 measurements. I do worry though that in places where the colorimetric devices are used for all there will be patients who are extubated due to the thought that they in fact have an esophageal intubation when the truth is they just need time to get the CO2 high enough to register a change in colour.
Anyways, this is food for thought and a chance to look at your own practice and see if it is in need of a tweak…

by All Things Neonatal | Mar 22, 2018 | Developmental care, kangaroo care, Neonatology, newborn
Skin to skin care or kangaroo care is all the rage and I am the first one to offer my support for it. Questions persist though as to whether from a physiological standpoint, babies are more stable in an isolette in a quiet environment or out in the open on their mother or father’s chests. Bornhorst et al expressed caution in their study Skin-to-skin (kangaroo) care, respiratory control, and thermoregulation. In a surprising finding, babies with an average gestational age of 29 weeks were monitored for a number of physiological parameters and found to have more frequent apnea and higher heart rates than when in an isolette. The study was small though and while there were statistical differences in these parameters they may not have had much clinical significance (1.5 to 2.8 per hour for apnea, bradycardia or desaturation events). Furthermore, does an increase in such events translate into any changes in cerebral oxygenation that might in turn have implications for later development? Tough to say based on a study of this magnitude but it certainly does raise some eyebrows.
What if we could look at cerebral oxygenation?
As you might have guessed, that is exactly what has been done by Lorenz L et al in their recent paper Cerebral oxygenation during skin-to-skin care in preterm infants not receiving respiratory support.The goal of this study was to look at 40 preterm infants without any respiratory distress and determine whether cerebral oxygenation (rStO2)was better in their isolette or in skin to skin care (SSC). They allowed each infant to serve as their own control by have three 90 minute periods each including the first thirty minutes as a washout period. Each infant started their monitoring in the isolette then went to SSC then back to the isolette. The primary outcome the power calculation was based on was the difference in rStO2 between SSC and in the isolette. Secondary measures looked at such outcomes as HR, O2 sat, active and quiet sleep percentages, bradycardic events as lastly periods of cerebral hypoxia or hyperoxia. Normal cerebral oxygenation was defined as being between 55 to 85%.
Surprising results?
Perhaps its the start of a trend but again the results were a bit surprising showing a better rStO2 when in the isolette (−1.3 (−2.2 to −0.4)%, p<0.01). Other results are summarized in the table below:
| Mean difference in outcomes |
| Variable |
SSC |
Isolette |
Difference in mean |
p |
| rStO2 |
73.6 |
74.8 |
-1.3 |
<0.01 |
| SpO2 (median) |
97 |
97 |
-1.1 |
0.02 |
| HR |
161 |
156 |
5 |
<0.01 |
| % time in quiet sleep |
58.6 |
34.6 |
24 |
<0.01 |
No differences were seen in bradycardic events, apnea, cerebral hypoexmia or hyperoxemia. The authors found that SSC periods in fact failed the “non-inferiority” testing indicating that from a rStO2 standpoint, babies were more stable when not doing SSC! Taking a closer look though one could argue that even if this is true does it really matter? What is the impact on a growing preterm infant if their cerebral oxygenation is 1.3 percentage points on average lower during SSC or if their HR is 5 beats per minute faster? I can’t help but think that this is an example of statistical significance without clinical significance. Nonetheless, if there isn’t a superiority of these parameters it does leave one asking “should we keep at it?”
Benefits of skin to skin care
Important outcomes such as reductions in mortality and improved breastfeeding rates cannot be ignored or the positive effects on family bonding that ensue. Some will argue though that the impacts on mortality certainly may be relevant in developing countries where resources are scarce but would we see the same benefits in developed nations. The authors did find a difference though in this study that I think benefits developing preterm infants across the board no matter which country you are in. That benefit is that of Quiet Sleep (QS). As preterm infants develop they tend to spend more time in QS compared to active sleep (AS). From Doussard- Roossevelt J, “Quiet sleep consists of periods of quiescence with regular respiration and heart rate, and synchronous EEG patterns. Active sleep consists of periods of movement with irregular respiration and heart rate, and desynchronous EEG patterns.” In the above table one sees that the percentage of time in QS was significantly increased compared to AS when in SSC. This is important as neurodevelopment is thought to advance during periods of QS as preterm infants age.
There may be little difference favouring less oxygen extraction during isolette times but maybe that isn’t such a good thing? Could it be that the small statistical difference in oxygen extraction is because the brain is more active in laying down tracks and making connections? Totally speculative on my part but all that extra quiet sleep has got to be good for something.
To answer the question of this post in the title I think the answer is a resounding yes for the more stable infant. What we don’t know at the moment except from anecdotal reports of babies doing better in SSC when really sick is whether on average critically ill babies will be better off in SSC. I suspect the answer is that some will and some won’t. While we like to keep things simple and have a one size fits all answer for most of our questions in the NICU, this one may not be so simple. For now I think we keep promoting SSC for even our sick patients but need to be honest with ourselves and when a patient just isn’t ready for the handling admit it and try again when more stable. For the more stable patient though I think giving more time for neurons to find other neurons and make new connections is a good thing to pursue!

by All Things Neonatal | Mar 14, 2018 | apnea of prematurity, caffeine, extubation, Neonatal, Neonatology, Prematurity, ventilation
Caffeine seems to be good for preterm infants. We know that it reduces the frequency of apnea in the this population and moreover facilitates weaning off the ventilator in a shorter time frame than if one never received it at all. The earlier you give it also seems to make a difference as shown in the Cochrane review on prophylactic caffeine. When given in such a fashion the chances of successful extubation increase. Less time on the ventilator not surprisingly leads to less chronic lung disease which is also a good thing.
I have written about caffeine more than once though so why is this post different? The question now seems to be how much caffeine is enough to get the best outcomes for our infants. Last month I wrote about the fact that as the half life of caffeine in the growing preterm infant shortens, our strategy in the NICU might be to change the dosing of caffeine as the patient ages. Some time ago though I wrote about the use of higher doses of caffeine and in the study analyzed warned that there had been a finding of increased cerebellar hemorrhage in the group randomized to receive the higher dosing. I don’t know about where you work but we are starting to see a trend towards using higher caffeine base dosing above 5 mg/kg/d. Essentially, we are at times “titrating to effect” with dosing being as high as 8-10 mg/kg/d of caffeine base.
Does it work to improve meaningful outcomes?
This month Vliegenthart R et al published a systematic review of all RCTs that compared a high vs low dosing strategy for caffeine in infants under 32 weeks at birth; High versus standard dose caffeine for apnoea: a systematic review. All told there were 6 studies that met the criteria for inclusion. Low dosing (all in caffeine base) was considered to be 5- 15 mg/kg with a maintenance dose of 2.5 mg/kg to 5 mg/kg. High dosing was a load of 5 mg/kg to 40 mg/kg with a maintenance of 2.5 mg/kg to 15 mg/kg. The variability in the dosing (some of which I would not consider high at all) makes the quality of the included studies questionable so a word of warning that the results may not truly be “high” vs “low” but rather “inconsistently high” vs. “inconsistently low”.
The results though may show some interesting findings that I think provide some reassurance that higher dosing can allow us to sleep at night.
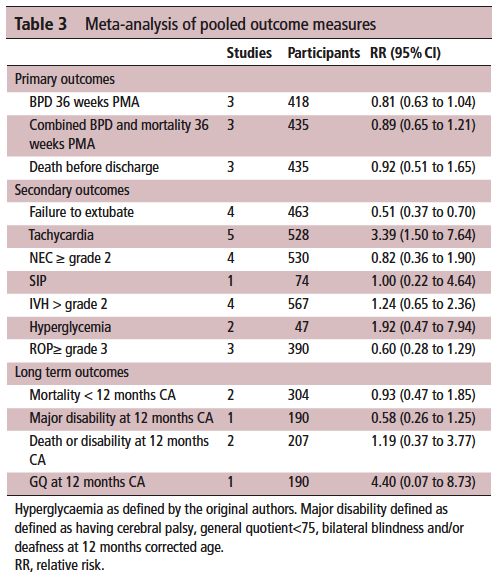
On the positive front, while there was no benefit to BPD and mortality at 36 weeks PMA they did find if they looked only at those babies who were treated with caffeine greater than 14 days there was a statistically significant difference in both reduction of BPD and decreased risk of BPD and mortality. This makes quite a bit of sense if you think about it for a moment. If we know that caffeine improves the chances of successful extubation and we also know it reduces apnea, then who might be on caffeine for less than 2 weeks? The most stable of babies I would expect! These babies were all < 32 weeks at birth. What the review suggests is that those babies who needed caffeine for longer durations benefit the most from the higher dose. I think I can buy that.
On the adverse event side, I suppose it shouldn’t surprise many that the risk of tachycardia was statistically increased with an RR of 3.4. Anyone who has explored higher dosing would certainly buy that as a side effect that we probably didn’t need an RCT to prove to us. Never mind that, have you ever taken your own pulse after a couple strong coffees in the morning?
What did it not show?
It’s what the study didn’t show that is almost equally interesting. The cerebellar hemorrhages seen in the study I previously wrote about were not seen at all in the other studies. There could be a lesson in there about taking too much stock in secondary outcomes in small studies…
Also of note, looking at longer term outcome measures there appears to be no evidence of harm when the patients are all pooled together. The total number of patients in all of these studies was 620 which for a neonatal systematic review is not bad. A larger RCT may be needed to conclusively tell us what to do with a high and low dosing strategy that we can all agree on. What do we do though in the here and now? More specifically, if you are on call tomorrow and a baby is on 5 mg/kg/d of caffeine already, will you intubate them if they are having copious apneic events or give them a higher dose of caffeine when CPAP or NIPPV that they are already on isn’t cutting it? That is where the truth about how you feel about the evidence really comes out. These decisions are never easy but unfortunately you sometimes have to make a decision and the perfect study hasn’t been done yet. I am not sure where you sit on this but I think this study while certainly flawed gives me some comfort that nothing is truly standing out especially given the fact that some of the “high dose” studies were truly high. Will see what happens with my next patient!
by All Things Neonatal | Jan 24, 2018 | caffeine, Neonatal, Neonatology, preemie, Prematurity
This has been a question that has befuddled Neonatologists for years. Get ten of us in a room and you will get a variety of responses ranging from (talking about caffeine base) 2.5 mg/kg/day to 10 mg/kg/day. We will espouse all of our reasons and question the issue of safety at higher doses but in the end do we really know? As I was speaking to a colleague in Calgary yesterday we talked about how convinced we are of our current management strategies but how we both recognize that half of what we think we know today we will be questioning in 10 years. So how convinced should we really be about caffeine?
Even the Cochrane Review Suggests There Is Something Amiss
Back in 2010 the Cochrane Collaboration examining 6 trials on caffeine for treating apnea of prematurity concluded “Methylxanthine is effective in reducing the number of apnoeic attacks and the use of mechanical ventilation in the two to seven days after starting treatment.” Notice the bolded section. Two to seven days. Interesting that we don’t see the effect last in perpetuity. Why might that be? Do babies become resistant with time or is there a change in the way these infants metabolize the drug such that levels in the bloodstream drop after that time point. It is almost certainly the latter and in the last 7 years have we really seen any response to this finding? I would say no for the most part although I don’t work in your unit so hard to say for sure. At least where I practice we pick a dose somewhere between 2.5-5 mg/kg/day and give a load of 10 mg/kg when we start the drug. From time to time we give a miniload of 5 mg/kg and may or may not increase the dose of maintenance based on the number of apneic events the babies are having. What if we could be proactive instead of reactive though. Do the babies need to have multiple events before we act or could we prevent the events from happening at all?
Proactive Treatment With Caffeine
We have known that caffeine clearance increases with postnatal age. The half-life of the drug shortens from about a week at the earliest gestational ages to 2-2.5 days by term equivalent age. For those infants who are older such as 32 weeks and above we expect them to be off caffeine (if they need it) within 2-3 weeks so I am not really talking about them but what about the babies born earlier than that or certainly MUCH earlier at 23 and 24 weeks who will be on caffeine possibly till term. Should one size (dose) fit all? No it really shouldn’t and some crafty researchers led by Koch G have published a paper that demonstrates why entitled Caffeine Citrate Dosing Adjustments to Assure Stable Caffeine Concentrations in Preterm Neonates.
In this paper the authors armed with knowledge of the half life of caffeine at different gestational ages were able to calculate the clearance of the drug at different postnatal ages to demonstrate in a model of a 28 week male infant weighing 1150g. The authors further took into account predicted weight changes and were able to calculate what the expected caffeine levels would be in the fictional infant at various time points. The target caffeine levels for this patient were a trough level of 15 -20 mg/L which are the currently acceptable ranges in the literature. The testing was first done using a standard load of 10 mg/kg (base) followed by 2.5 mg/kg/d (base) and demonstrated levels which yielded the following graph over time. 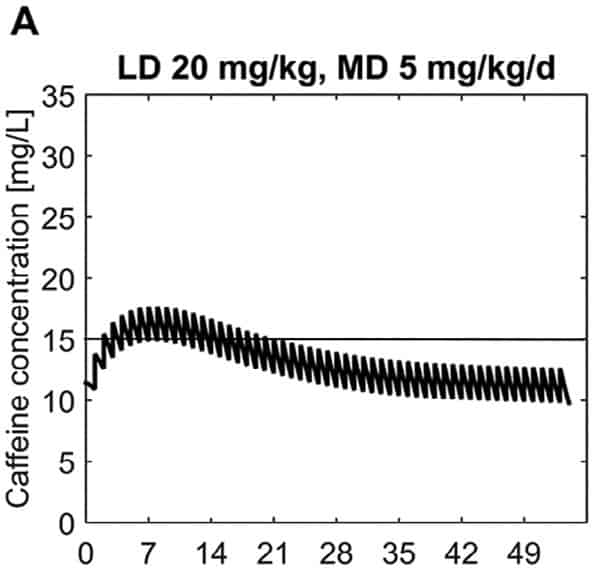 What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next.
What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next. 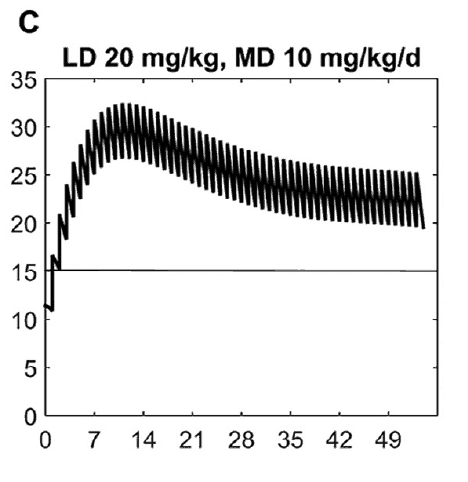 Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve.
Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve.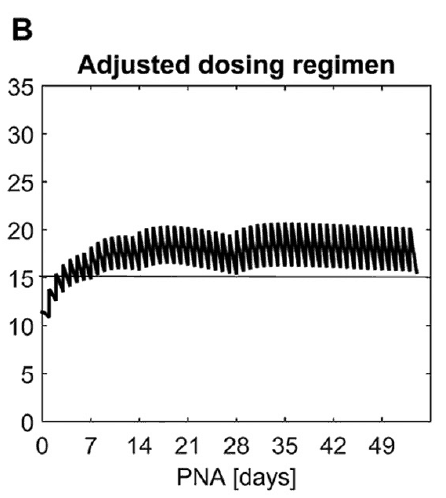 As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.
As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.
A Suggestion For The Future
What grabbed my attention here is the possibility that we could take a proactive rather than reactive approach to these infants. Once a small baby is controlled on their dose of caffeine whether it is 2.5, 3, 5 or even 6 mg/kg/d of caffeine should we wait for more events to occur and then react by increasing caffeine? What if we are too late to respond and the patient is intubated. What effect does this have on the developing lung, what about the brain that is subjected to bradycardic events with resultant drops in cardiac output and cerebral perfusion. Perhaps the solution is to work with our pharmacists and plan to increase dosing at several time points in the infants journey through the NICU even if they aren’t showing symptoms yet. No doubt this is a change in approach at least for the unit I work in but one that should start with a conversation!
by All Things Neonatal | Jan 15, 2018 | BPD, Neonatal, Neonatology, newborn, outcome, preemie, Prematurity, steroids
This must be one of my favourite topics as I have been following the story of early hydrocortisone to reduce BPD for quite some time. It becomes even more enticing when I have met the authors of the studies previously and can see how passionate they are about the possibilities. The PREMILOC study was covered on my site twice now, with the first post being A Shocking Change in Position. Postnatal steroids for ALL microprems? and the second reviewing the 22 month outcome afterwards /2017/05/07/early-hydrocortisone-short-term-gain-without-long-term-pain/.
The intervention here was that within 24 hours of birth babies born between 24-27 weeks gestational age were randomized to receive placebo or hydrocortisone 1 mg/kg/d divided q12h for one week followed by 0.5 mg/kg/d for three days. The primary outcome was rate of survival without BPD at 36 weeks PMA. The finding was a positive one with a 9% reduction in this outcome with the use of this strategy. Following these results were the two year follow-up which reported no evidence of harm but the planned analysis by gestational age groupings of 24-25 and 26-27 weeks was not reported at that time but it has just been released this month.
Is there a benefit?
Of the original cohort the authors are to be commended here as they were able to follow-up 93% of all infants studied at a mean age of 22 months. The methods of assessing their neurological status have been discussed previously but essentially comprised standardized questionnaires for parents, assessment tools and physical examinations.
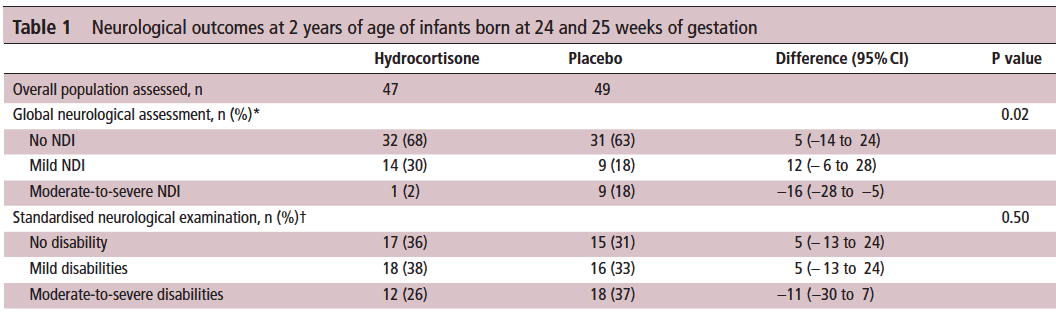
Let’s start off with what they didn’t find. There was no difference between those who received placebo vs hydrocortisone in the 26-27 week group but where it perhaps matters most there was. The infants born at 24-25 weeks are certainly some of our highest risk infants in the NICU. It is in this group that the use of hydrocortisone translated into a statistically significant reduction in the rate of neurodevelopmental impairment. The Global Neurological Assessement scores demonstrated a significant improvement in the hydrocortisone group with a p value of 0.02. Specifically moderate to severe disability was noted in 18% compared to 2% in the group receiving hydrocortisone.They did not find a difference in the neurological exam but that may reflect the lack of physical abnormalities with cognitive deficit remaining. It could also be explained perhaps by the physical examination not being sensitive enough to capture subtle differences.
Why might this be?
Adding an anti-inflammatory agent into the early phase of a preemies life might spare the brain from white matter damage. Inflammation is well known to inflict injury upon the developing brain and other organs (think BPD, ROP) so dampening these factors in the first ten days of life could bring about such results via a mechanism such as that. When you look at the original findings of the study though, a couple other factors also pop up that likely contribute to these findings as well. Infants in the hydrocortisone group had a statistical reduction in the rate of BPD and PDA ligations. Both of these outcomes have been independently linked to adverse neurodevelopmental outcome so it stands to reason that reducing each of these outcomes in the most vulnerable infants could have a benefit.
In fact when you add everything up, is there much reason not to try this approach? Ten days of hydrocortisone has now been shown to reduce BPD, decrease PDA ligations and importantly in the most vulnerable of our infants improve their developmental outcome. I think with this information at our fingertips it becomes increasingly difficult to ignore this approach. Do I think this will become adopted widely? I suspect there will be those who take the Cochrane approach to this and will ask for more well designed RCTs to be done in order to replicate these results or at least confirm a direction of effect which can then be studied as part of a systematic review. There will be those early adopters though who may well take this on. It will be interesting to see as these centres in turn report their before and after comparisons in the literature what the real world impact of this approach might be.
Stay tuned as I am sure this is not the last we will hear on this topic!






 What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next.
What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next.  Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve.
Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve. As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.
As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.
